Characteristics and Trends in HIV Testing Among Medicaid Enrollees Diagnosed as Having Schizophrenia
Abstract
Objective:
People with schizophrenia have more HIV risk factors and higher rates of HIV infection than the general U.S. population. The authors aimed to examine HIV testing patterns in this population nationally and by demographic characteristics and presence of high-risk comorbid conditions.
Methods:
This retrospective longitudinal study compared HIV testing between Medicaid-only enrollees with schizophrenia and without schizophrenia during 2002–2012 (N=6,849,351). Interrupted time series were used to analyze the impacts of the 2006 federal policy change recommending expanded HIV testing. Among enrollees with schizophrenia, multivariable logistic regression was used to estimate associations between testing and both demographic characteristics and comorbid conditions. Sensitivity analyses were also conducted.
Results:
Enrollees diagnosed as having schizophrenia had consistently higher HIV testing rates than those without schizophrenia. When those with comorbid substance use disorders or sexually transmitted infections were excluded, testing was higher for individuals without schizophrenia (p<0.001). The federal policy change likely increased testing for both groups (p<0.001), but the net change was greater for those without schizophrenia (3.1 vs. 2.2 percentage points). Among enrollees with schizophrenia, testing rates doubled during 2002–2012 (3.9% to 7.2%), varied across states (range 17 percentage points), and tripled for those with at least one annual nonpsychiatric medical visit (vs. no visit; adjusted OR=3.10, 95% CI=2.99–3.22).
Conclusions:
Nationally, <10% of enrollees with schizophrenia had annual HIV testing. Increases appear to be driven by high-risk comorbid conditions and nonpsychiatric encounters, rather than by efforts to target people with schizophrenia. Psychiatric guidelines for schizophrenia care should consider HIV testing alongside annual metabolic screening.
HIGHLIGHTS
HIV testing rates for people diagnosed as having schizophrenia were consistently higher than for individuals without schizophrenia.
For both groups, HIV testing increased from 2002 to 2012, with a significant upturn (p<0.001) after policy changes from the Centers for Disease Control and Prevention in 2006; however, the net increase in testing was lower for people with schizophrenia.
Among individuals diagnosed as having schizophrenia, the likelihoods of HIV testing for those who had at least one annual nonpsychiatric outpatient medical visit were more than double (in 2002) and triple (in 2012) those with no annual visit.
Although HIV testing increased from 2002 to 2012 for individuals diagnosed as having schizophrenia, >90% of this population was still not tested for HIV, and little evidence of focused testing efforts was observed for this group, with documented elevations in risk for HIV infection.
People with schizophrenia spectrum and bipolar disorders have HIV and AIDS at higher rates than the general U.S. population (see the online supplement to this article) (1). In the U.S. population, HIV prevalence was 0.6% (95% CI=0.5%–0.8%) in 2011 (2), and a meta-analysis of U.S. studies published between 1980 and 2015 reported that people with severe mental illness have a pooled HIV prevalence of 6.0% (95% CI=4.3%–8.3%) (1). In comparison, the most recent multisite cross-sectional study, conducted from 2009 to 2011, reported an HIV prevalence of 4.8% (3) among people with schizophrenia.
The prevalence of HIV risk factors in the United States is also higher among people with schizophrenia spectrum and bipolar disorders (4), yet many characteristics of this population that increase risk for HIV infection are present also in the general population (e.g., injection drug use and condomless sex) (5). Adverse social and systemic factors can also increase vulnerability to HIV (e.g., sexual violence and unstable housing) (6). Since the 1990s, repeated calls for improved prevention, identification, and treatment have been made to reduce HIV-related morbidity, mortality, and transmission rates (7, 8). These suggestions include HIV testing efforts in community mental health settings (9) and research expansion by NIMH to drive responsive policy (10).
Research on HIV testing among people with schizophrenia is limited. A review of U.S. research published through 2007 found that 1-year HIV testing rates for people with schizophrenia spectrum and bipolar disorders ranged from 17% to 47%, with lifetime testing rates ranging from 11% to 89% (11). However, the heterogeneities of study settings, samples, methods, and definitions across publications have impeded detailed interpretation of this wide variation (11) (see the online supplement). Two more recent studies reported 1-year testing rates of 6.7% and 7.2% (12, 13). Notably, none of the previous studies included a national cohort with enough power to estimate geographic differences or to help identify characteristics that may influence HIV testing.
In this study, we aimed to fill these research gaps by examining HIV testing among people with schizophrenia, compared with a frequency-matched control group, with Medicaid claims records drawn from a large, national, retrospective longitudinal cohort. Considering the prevalence of Medicaid recipients with schizophrenia (14), Medicaid claims enable examination of HIV testing rates across multiple settings and multilevel predictors of HIV testing during a substantial observation window, with sufficient statistical power for subgroup analysis. Although national Medicaid data do not become generally available for research until years after collection, they offer detailed insights into national trends, including U.S. HIV testing patterns among people with schizophrenia and the impact of individual, institutional, and policy-related factors.
Methods
Study Cohort
This retrospective observational study used national administrative Medicaid Analytic eXtract (MAX) data from the Centers for Medicare and Medicaid Services from January 1, 2001, to December 31, 2012. Medicaid is a major payer of care for those with schizophrenia, and the largest among people with HIV in the United States (14–16). We compared billing claims for HIV testing among enrollees diagnosed as having schizophrenia with claims among people without schizophrenia or similar conditions (total N=6,849,351). Although we recognize that these were clinician-reported diagnoses with uncertain validity, we use the term “people (or individuals or enrollees) with schizophrenia” throughout this article. Eligibility criteria included being a Medicaid enrollee ages 15–64 years as of December 31 during the observed calendar year, having at least 11 months of eligibility during the calendar year, and residence in one of the 45 U.S. states with available Medicaid data during the study period. Data were deidentified; therefore, informed consent was waived and expediated approval was provided by the University of California, San Francisco Institutional Review Board (17–21998).
Exposure Assessment
Individuals were determined to have schizophrenia if they had at least one inpatient claim or at least two outpatient claims for schizophrenia (ICD-9-CM 295.x) within any given 6-month period during the observed calendar year (17). To avoid selecting a comparison group that differed dramatically on strong confounders of HIV testing for participants with schizophrenia, we selected a frequency-matched control group, within each year of first eligibility for the schizophrenia cohort, that had the same distributions of age, sex, and race-ethnicity as the schizophrenia group.
Individuals in the control group were excluded starting on the date of any observed claim for schizophrenia. We also excluded all persons in the control group with a diagnosis of bipolar disorder, psychosis (not otherwise specified), delusional disorders, and pervasive developmental disorders at any point during the study period. The exclusion of individuals with these disabling mental health conditions created a control group that contrasted sharply with the cohort with schizophrenia.
Outcome Assessment
Participants with HIV infection (ICD-9-CM codes 042, 079.53, V08, and 795.71) documented ≥ 12 months before the calendar year were excluded. For example, individuals with newly identified HIV infection in 2008 were excluded from subsequent years (2009–2012). Those who were screened in the previous calendar year, but were not diagnosed as having HIV, were carried forward.
HIV testing was assessed dichotomously (0, untested; 1, tested) in every year of the study period by using CPT codes (see the online supplement). If an individual test was billed, we also collected information about the place of service to determine whether the test occurred in a community mental health setting.
Covariates
All individual characteristics were coded categorically. Demographic covariates included age (15–19, 20–29, 30–39, 40–49, 50–59, 60–64 years), sex (male, female), and race-ethnicity (White, Black, Asian or Pacific Islander, Hispanic/Latino, Native Hawaiian or other Pacific Islander, American Indian/Alaska Native, multiracial, unknown). Co-occurring clinical diagnoses included substance use disorders (alcohol, opioids, cocaine, amphetamine, cannabis, or other) and selected other mental and general medical disorders (e.g., depression or hypertension). On the basis of existing evidence that nonpsychiatric outpatient medical visits increase the odds of HIV testing for people with schizophrenia (13), we included these visits as a covariate. We included specific medical conditions with a linkage to risks for HIV infection, increased probability of testing, or both, including hepatitis B virus (HBV), hepatitis C virus (HCV), and sexually transmitted infections (STIs) (i.e., herpes simplex virus [HSV], chlamydia, syphilis, and gonococcal infection). We also included metabolic disorders that commonly affect people with schizophrenia, including diabetes mellitus, hypertension, and dyslipidemia. We assessed clustered co-occurring clinical diagnoses (any substance use disorder, any STI, and any metabolic disorder diagnosis) dichotomously (0, none; 1, yes). (A list of ICD-9-CM diagnostic codes is available in the online supplement.)
Statistical Analysis
After matching, enrollees dually eligible for Medicare were excluded to avoid assessment of HIV testing covered by Medicare. Frequencies described individual characteristics, and chi-square tests of homogeneity were used to evaluate differences between people with schizophrenia and individuals in the control group. All estimates were adjusted for frequency-matching factors (age, sex, and race-ethnicity). Distributions of adjusted HIV testing were assessed over time by cohort. Adjusted state-level HIV testing for 2012 and the change in testing between 2002 and 2012 were mapped. Adjusted logistic regression was used to estimate associations of HIV testing with state, year, and individual characteristics. Two-sided associations were estimated by using robust standard errors to account for individuals being followed up over time. We also estimated interactions between schizophrenia and other individual characteristics. Because the Centers for Disease Control and Prevention (CDC) updated HIV testing guidelines in 2006 to recommend at least annual testing for individuals at higher risk (8), we assessed whether and to what extent trends in testing changed both for individuals with schizophrenia and for individuals without schizophrenia by using a comparative interrupted time-series analysis from 2002 through 2012, adjusted for covariates. All statistical analyses were performed with Stata, version 16.
Sensitivity Analysis
Nonpsychiatric outpatient medical visits were conceptualized as possible mediators between study cohort characteristics and receiving an HIV test, given that care for people with schizophrenia may differ from care for individuals in the control group (18). If true, overcontrolling for nonpsychiatric medical visits could bias estimates toward the null, because we expected these visits to increase the likelihood of being tested. To test this assumption, we conducted a sensitivity analysis, adjusting for utilization of outpatient nonpsychiatric medical visits (a list of CPT codes is available in the online supplement). In addition, we assessed the potential influence of having comorbid conditions that increase HIV risk (such as substance use disorders and STIs) on HIV testing between individuals with and those without schizophrenia by using adjusted nested regression and by restricting the cohort to those without the comorbid condition.
Results
HIV Testing, by Schizophrenia or Control Group (2002–2012)
Throughout the 10-year study period examined, overall HIV testing approximately doubled nationally, from 3.8% in 2002 to 7.5% in 2012 (see the online supplement). HIV testing rates were consistently higher for people with schizophrenia than for individuals in the control group across all years (p<0.001). Among those with schizophrenia, HIV testing increased from 3.9% (95% CI=3.8%–4.0%) in 2002 to 7.2% (95% CI=7.1%–7.2%) in 2012, with an adjusted increase of 3.7 percentage points (95% CI=3.6–3.8) over this period. Among individuals in the control group, HIV testing increased from 2.9% (95% CI=2.9%–3.0%) in 2002 to 5.8% (95% CI=5.8%–5.9%) in 2012, with an adjusted increase of 3.8 percentage points (95% CI=3.7–3.9). After the 2006 change in CDC HIV testing guidelines (8), trends in testing shifted upward for both groups (p<0.001); however, the net change was higher for the control group (3.1 percentage points, 95% CI=2.9–3.2) than for people with schizophrenia (2.2 percentage points, 95% CI=2.0–2.4) (Figure 1).
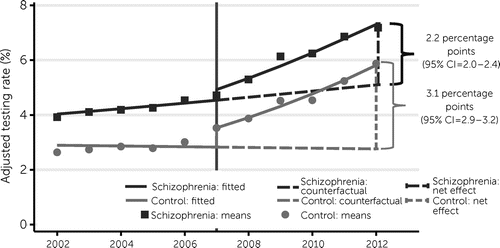
FIGURE 1. Adjusted interrupted time-series analysis of HIV testing among Medicaid-only enrollees with or without schizophrenia, 2002–2012 (N=6,849,351)
State-Level HIV Testing in the Population With Schizophrenia
As depicted in Figure 2, state-level HIV testing among individuals with schizophrenia in 2012 (N=368,274) ranged from 0.4% to 17.2%, after data were adjusted for age, sex, and race-ethnicity. (Full state estimates are reported in the online supplement.) Changes in HIV testing from 2002 to 2012 ranged by state from −2.2 to 10.9 percentage points among people with schizophrenia (see the online supplement).
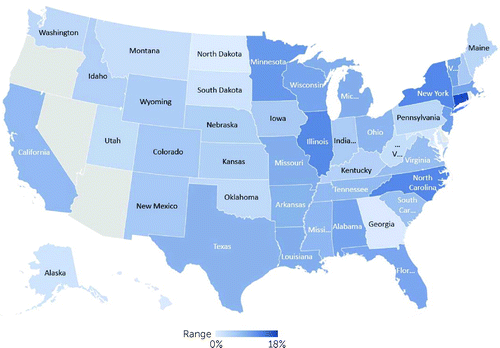
FIGURE 2. HIV testing rates among Medicaid-only enrollees with schizophrenia, adjusted for age, sex, and race-ethnicity, 2012 (N=368,274)
Study Cohort Characteristics, 2002 and 2012
Individual characteristics at the beginning and end of the study period are presented in Table 1. The entire cohort comprised 538,572 individuals in 2002 and 854,589 individuals in 2012. The cohort with schizophrenia was diverse in terms of age, gender, and race-ethnicity, with 235,066 individuals in 2002 and 368,274 in 2012.
| 2002 (N=538,572) | 2012 (N=854,589) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Schizophrenia (N=235,066) | Control (N=303,506) | Schizophrenia (N=368,274) | Control (N=486,315) | |||||||
| Characteristic | N | % | N | % | p | N | % | N | % | pa |
| Sex | <.001 | .006 | ||||||||
| Female | 119,770 | 51.0 | 151,714 | 50.0 | 173,062 | 47.0 | 227,063 | 46.7 | ||
| Male | 115,296 | 49.0 | 151,792 | 50.0 | 195,212 | 53.0 | 259,252 | 53.3 | ||
| Race-ethnicity | <.001 | <.001 | ||||||||
| White | 103,390 | 44.0 | 157,842 | 52.0 | 142,497 | 38.7 | 216,084 | 44.4 | ||
| Black | 78,577 | 33.4 | 90,786 | 29.9 | 132,746 | 36.0 | 159,609 | 32.8 | ||
| American Indian or Alaska Native | 1,683 | 0.7 | 2,219 | 0.7 | 3,108 | 0.8 | 4,239 | 0.9 | ||
| Asian or Pacific Islander | 3,832 | 1.6 | 6,299 | 2.1 | 7,360 | 2.0 | 11,267 | 2.3 | ||
| Hispanic/Latino | 20,572 | 8.8 | 25,554 | 8.4 | 40,554 | 11.0 | 50,493 | 10.4 | ||
| Native Hawaiian or other Pacific Islander | 4,011 | 1.7 | 3,437 | 1.1 | 5,310 | 1.4 | 4,620 | 1.0 | ||
| Multiracial | 437 | 0.2 | 220 | 0.1 | 1,297 | 0.4 | 1,610 | 0.3 | ||
| Unknown | 22,564 | 9.6 | 17,149 | 5.7 | 35,402 | 9.6 | 38,393 | 7.9 | ||
| Age in years | <.001 | <.001 | ||||||||
| 15–19 | 8,289 | 3.5 | 8,705 | 2.9 | 14,421 | 3.9 | 27,634 | 5.7 | ||
| 20–29 | 32,261 | 13.7 | 40,092 | 13.2 | 62,417 | 16.9 | 77,458 | 15.9 | ||
| 30–39 | 48,861 | 20.8 | 71,428 | 23.5 | 67,709 | 18.4 | 100,169 | 20.6 | ||
| 40–49 | 76,187 | 32.4 | 101,339 | 33.4 | 83,140 | 22.6 | 116,640 | 24.0 | ||
| 50–59 | 53,180 | 22.6 | 63,100 | 20.8 | 106,191 | 28.8 | 125,155 | 25.7 | ||
| 60–64 | 16,288 | 6.9 | 18,842 | 6.2 | 34,396 | 9.3 | 39,259 | 8.1 | ||
| Comorbid conditionb | ||||||||||
| Substance use | ||||||||||
| Alcohol | 21,844 | 9.3 | 9,239 | 3.0 | <.001 | 38,972 | 10.6 | 14,351 | 3.0 | <.001 |
| Opioid | 5,520 | 2.3 | 4,053 | 1.3 | <.001 | 15,643 | 4.2 | 9,738 | 2.0 | <.001 |
| Cocaine | 10,938 | 4.7 | 2,769 | 0.9 | <.001 | 18,622 | 5.1 | 3,786 | 0.8 | <.001 |
| Amphetamine | 1,566 | 0.7 | 340 | 0.1 | <.001 | 5,162 | 1.4 | 796 | 0.2 | <.001 |
| Cannabis | 7,461 | 3.2 | 1,363 | 0.4 | <.001 | 23,784 | 6.5 | 4,730 | 1.0 | <.001 |
| Other | 16,063 | 6.8 | 4,323 | 1.4 | <.001 | 34,119 | 9.3 | 9,289 | 1.9 | <.001 |
| Any substance use | 38,634 | 16.4 | 15,933 | 5.2 | <.001 | 82,035 | 22.3 | 30,838 | 6.3 | <.001 |
| No substance use (reference) | 196,432 | 83.6 | 287,573 | 94.8 | 286,239 | 77.7 | 455,477 | 93.7 | ||
| Psychiatric | ||||||||||
| Anxiety | 20,042 | 8.5 | 13,963 | 4.6 | <.001 | 57,705 | 15.7 | 32,786 | 6.7 | <.001 |
| Depression | 62,923 | 26.8 | 27,681 | 9.1 | <.001 | 104,851 | 28.5 | 45,367 | 9.3 | <.001 |
| General medical | ||||||||||
| Diabetes mellitus | 39,656 | 16.9 | 32,118 | 10.6 | <.001 | 80,464 | 21.8 | 58,421 | 12.0 | <.001 |
| Hypertension | 63,125 | 26.9 | 59,345 | 19.6 | <.001 | 132,679 | 36.0 | 114,492 | 23.5 | <.001 |
| Dyslipidemia | 37,235 | 15.8 | 33,023 | 10.9 | <.001 | 92,812 | 25.2 | 76,836 | 15.8 | <.001 |
| Metabolic disorder | 94,951 | 40.4 | 83,552 | 27.5 | <.001 | 184,166 | 50.0 | 154,049 | 31.7 | <.001 |
| No metabolic disorder (reference) | 140,115 | 59.6 | 219,954 | 72.5 | 184,108 | 50.0 | 332,266 | 68.3 | ||
| Hepatitis B | 1,455 | 0.6 | 957 | 0.3 | <.001 | 2,137 | 0.6 | 1,711 | 0.4 | <.001 |
| Hepatitis C | 6,951 | 3.0 | 5,378 | 1.8 | <.001 | 15,200 | 4.1 | 8,402 | 1.7 | <.001 |
| Sexually transmitted infection | ||||||||||
| Herpes simplex | 1,532 | 0.7 | 1,453 | 0.5 | <.001 | 4,063 | 1.1 | 3,475 | 0.7 | <.001 |
| Chlamydia | 391 | 0.2 | 594 | 0.2 | .012 | 962 | 0.3 | 1,520 | 0.3 | <.001 |
| Syphilis | 837 | 0.4 | 499 | 0.2 | <.001 | 1,084 | 0.3 | 719 | 0.1 | <.001 |
| Gonococcal infection | 523 | 0.2 | 534 | 0.2 | <.001 | 669 | 0.2 | 735 | 0.2 | .001 |
| Any | 3,115 | 1.3 | 2,839 | 0.9 | <.001 | 6,404 | 1.7 | 6,161 | 1.3 | <.001 |
| None (reference) | 231,951 | 98.7 | 300,667 | 99.1 | 361,870 | 98.3 | 480,154 | 98.7 | ||
TABLE 1. Characteristics of Medicaid-only enrollees with or without schizophrenia, 2002 and 2012 (N=1,393,161)
Adjusted HIV Testing in the Population With Schizophrenia, 2002 and 2012
Table 2 presents HIV testing estimates in 2002 and 2012 among enrollees with schizophrenia, adjusted for age, sex, race-ethnicity, and comorbid conditions. In 2002, testing rates were highest for individuals who were Black, Hispanic/Latino, female, or ages 15–39 years or who had comorbid cocaine and other drug use and certain comorbid medical diagnoses, specifically STIs such as HCV, HSV, and syphilis infections. In 2012, HIV testing was again highest for these groups. Increases in testing from 2002 to 2012 were statistically significant for almost all demographic characteristics and comorbid conditions (p<0.001 for most estimates).
| 2002 (N=235,066) | 2012 (N=368,274) | Change in testing (2012 vs. 2002) | |||||
|---|---|---|---|---|---|---|---|
| Characteristic | Adjusted % | 95% CI | Adjusted % | 95% CI | Adjusted percentage points | 95% CI | pa |
| Sex | |||||||
| Female | 6.0 | 5.9, 6.2 | 10.1 | 9.9, 10.2 | 3.9 | 3.7, 4.1 | <.001 |
| Male | 3.9 | 3.8, 4.1 | 6.3 | 6.2, 6.4 | 2.5 | 2.3, 2.6 | <.001 |
| Race-ethnicity | |||||||
| Black | 5.6 | 5.5, 5.8 | 10.8 | 10.6, 11.0 | 5.4 | 5.2, 5.6 | <.001 |
| American Indian or Alaska Native | 3.7 | 2.8, 4.5 | 5.3 | 4.6, 6.1 | 1.7 | .5, 3.0 | .006 |
| Asian or Pacific Islander | 1.8 | 1.4, 2.3 | 6.0 | 5.4, 6.5 | 3.8 | 3.2, 4.5 | <.001 |
| Hispanic/Latino | 6.2 | 5.9, 6.5 | 10.3 | 10.0, 10.6 | 4.3 | 3.8, 4.7 | <.001 |
| Native Hawaiian or other Pacific Islander | 4.1 | 3.4, 4.7 | 4.5 | 3.9, 5.1 | .5 | −.3, 1.2 | .242 |
| Multiracial | 3.8 | 2.1, 5.5 | 9.9 | 8.3, 11.4 | 6.8 | 4.2, 9.4 | <.001 |
| Unknown | 4.8 | 4.5, 5.1 | 7.0 | 6.7, 7.2 | 2.1 | 1.7, 2.4 | <.001 |
| White | 4.2 | 4.0, 4.3 | 5.6 | 5.5, 5.7 | 1.3 | 1.2, 1.5 | <.001 |
| Age in years | |||||||
| 15–19 | 7.2 | 6.5, 7.8 | 10.6 | 10.1, 11.1 | 3.4 | 2.7, 4.1 | <.001 |
| 20–29 | 7.8 | 7.4, 8.1 | 12.2 | 12.0, 12.5 | 4.4 | 4.1, 4.8 | <.001 |
| 30–39 | 6.4 | 6.1, 6.6 | 9.8 | 9.6, 10.0 | 3.4 | 3.1, 3.7 | <.001 |
| 40–49 | 5.0 | 4.8, 5.2 | 7.9 | 7.8, 8.1 | 3.1 | 2.8, 3.3 | <.001 |
| 50–59 | 3.2 | 3.0, 3.3 | 5.9 | 5.7, 6.0 | 2.7 | 2.5, 2.9 | <.001 |
| 60–64 | 1.9 | 1.6, 2.1 | 4.0 | 3.8, 4.2 | 2.0 | 1.7, 2.3 | <.001 |
| Comorbid condition | |||||||
| Substance use | |||||||
| Alcohol | 5.8 | 5.4, 6.1 | 9.7 | 9.4, 10.0 | 4.9 | 4.4, 5.4 | <.001 |
| Opioid | 5.6 | 5.1, 6.1 | 9.6 | 9.2, 10.1 | 5.3 | 4.3, 6.4 | <.001 |
| Cocaine | 6.2 | 5.7, 6.6 | 10.2 | 9.7, 10.6 | 6.9 | 6.1, 7.8 | <.001 |
| Amphetamine | 7.2 | 6.1, 8.2 | 8.6 | 8.0, 9.3 | .3 | −1.5, 2.2 | .731 |
| Cannabis | 4.6 | 4.2, 5.0 | 8.8 | 8.5, 9.2 | 6.3 | 5.6, 7.1 | <.001 |
| Other drug | 7.0 | 6.6, 7.3 | 10.8 | 10.4, 11.1 | 5.0 | 4.3, 5.6 | <.001 |
| Psychiatric | |||||||
| Anxiety | 6.1 | 5.8, 6.4 | 8.9 | 8.7, 9.1 | 2.7 | 2.3, 3.2 | <.001 |
| Depression | 5.8 | 5.7, 6.0 | 9.3 | 9.1, 9.5 | 3.8 | 3.5, 4.1 | <.001 |
| General medical | |||||||
| Hepatitis B | 7.8 | 6.7, 9.0 | 14.5 | 13.1, 16.0 | 8.1 | 5.5, 10.7 | <.001 |
| Hepatitis C | 13.6 | 12.7, 14.5 | 17.5 | 16.8, 18.1 | 4.0 | 3.0, 5.1 | <.001 |
| Diabetes mellitus | 4.8 | 4.6, 5.0 | 8.1 | 7.9, 8.3 | 3.3 | 3.0, 3.6 | <.001 |
| Hypertension | 5.3 | 5.1, 5.5 | 8.5 | 8.4, 8.7 | 3.3 | 3.1, 3.5 | <.001 |
| Dyslipidemia | 6.4 | 6.1, 6.7 | 9.5 | 9.3, 9.7 | 2.8 | 2.5, 3.1 | <.001 |
| Sexually transmitted infection | |||||||
| Herpes simplex | 13.8 | 12.2, 15.3 | 21.4 | 20.2, 22.6 | 9.3 | 6.8, 11.8 | <.001 |
| Chlamydia | 11.9 | 8.9, 14.8 | 26.6 | 23.8, 29.3 | 22.7 | 17.3, 28.1 | <.001 |
| Syphilis | 15.2 | 12.9, 17.5 | 20.9 | 18.6, 23.2 | 8.3 | 4.3, 12.3 | <.001 |
| Gonococcal infection | 10.3 | 7.9, 12.7 | 22.0 | 18.9, 25.1 | 20.1 | 14.7, 25.4 | <.001 |
TABLE 2. Estimated HIV testing rates among Medicaid-only recipients with schizophrenia, adjusted for age, sex, race-ethnicity, and comorbid conditions, 2002 and 2012 (N=603,340)
Multivariable Logistic Regression
Adjusted ORs (AORs) for HIV testing among people with schizophrenia varied by individual characteristics in 2002 and 2012 (see the online supplement). In Figure 3, 2012 estimates are presented by demographic factors and diagnosis of comorbid conditions. The likelihood of HIV testing was consistently higher among females (compared with males) and among individuals who were Black, Hispanic, multiracial, or of unknown race (vs. White individuals); younger (vs. those ages 60–64); or had a substance use disorder, comorbid medical condition, or STI (vs. those without such conditions). We noted five exceptions: HIV testing was lower for people who were Native Hawaiians or other Pacific Islanders (vs. White; AOR=0.79, 95% CI=0.68–0.91), and we observed no significant differences for individuals who were American Indian/Alaska Native (vs. White; AOR=0.95, 95% CI=0.81–1.11) or Asian or Pacific Islanders (vs. White; AOR=1.08, 95% CI=0.97–1.20) or who had diagnoses of comorbid amphetamine use (vs. no use; AOR=1.08, 95% CI=0.98–1.18) or diabetes (vs. no diabetes; AOR=1.00, 95% CI=0.96–1.03). The likelihood of HIV testing was at least triple for those ages 15–29 (vs. ages 60–64) and for those with any diagnosed STI (vs. no STI), with the highest likelihood among those with comorbid chlamydia (AOR=4.64, 95% CI=3.97–5.42).
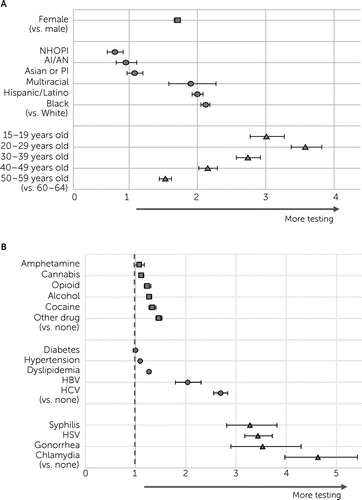
FIGURE 3. Adjusted ORs for HIV testing among Medicaid-only enrollees with schizophrenia, 2012 (N=368,274)a
aA: demographic characteristics. B: comorbid conditions. Error bars indicate 95% CIs. AI/AN, American Indian or Alaska Native; HBV, hepatitis B virus; HCV, hepatitis C virus; HSV, herpes simplex virus; NHOPI, Native Hawaiian or other Pacific Islander; PI, Pacific Islander.
Further adjustment for having at least one annual nonpsychiatric outpatient medical visit attenuated most 2012 estimates (by 3.7%–18.2%) or resulted in no change (see the online supplement). The greatest changes in attenuated estimates were among people with schizophrenia and comorbid HBV (15%) or hypertension (18%). Among people diagnosed as having schizophrenia, the likelihood of HIV testing for those who had at least one annual nonpsychiatric outpatient medical visit was more than double in 2002 (AOR=2.69, 95% CI=2.55–2.83) and triple in 2012 (AOR=3.10, 95% CI=2.99–3.22) compared with those with no annual visit. Notably, only one HIV test claim occurred in a community mental health clinic during 2002, and none occurred in this clinic during 2012.
Having a substance use disorder or STI influenced the likelihood of HIV testing for people with schizophrenia, compared with the control group (see the online supplement). In nested models, no statistically significant difference was found in testing among those with versus those without schizophrenia after we accounted for comorbid STI. In models restricted to those with no substance use disorder or STI, testing rates were consistently lower for people with schizophrenia compared with enrollees in the control group (p<0.001).
Discussion
In this national, longitudinal study of Medicaid-only enrollees with schizophrenia, adjusted HIV testing rates were <10% annually. Although adjusted HIV testing rates increased from 3.9% to 7.2% during the 2002–2012 period, the increases appeared to be primarily driven by the presence of medical conditions known to be associated with HIV (e.g., STIs, hepatitis, and substance use disorders), rather than a targeted policy focus to increase HIV testing for people with schizophrenia.
People with schizophrenia benefited from the 2006 CDC policy change in HIV testing guidelines. However, the impact of this policy change was greater for individuals in the control group than for individuals with schizophrenia (3.1 vs. 2.2 percentage-point increase in testing). In models that excluded individuals with a substance use disorder or STI, testing rates were consistently lower for people with schizophrenia than for individuals in the control group, an important finding given that advocates hoped that the 2006 policy would increase routine HIV testing practices (19). Variation among states in the adoption of the CDC’s recommendations may partially explain the state-level differences in HIV testing we observed.
Our findings offer empirical evidence that nonpsychiatric outpatient medical visits likely increase the probability of HIV testing among people with schizophrenia; those with more nonpsychiatric outpatient medical visits had higher odds of receiving HIV testing. Given this finding, state Medicaid and public mental health authorities could consider incentives for integration of care to improve HIV testing among people diagnosed as having schizophrenia. Taken together, our results suggest that HIV testing guidelines for the general population provide some benefit to people with schizophrenia, especially those with comorbid conditions. Our results align with those of studies suggesting that providers may be responding to patient risk factors unrelated to schizophrenia (11). As such, there is a need for interventions to promote HIV testing for individuals with schizophrenia who do not have documented comorbid conditions, particularly in mental health settings.
Although our data were >10 years old, our findings are critically important and have major policy implications. First, in 2017, the CDC undertook a literature review to evaluate their 2006 recommendations for HIV screening of high-risk populations and decided to leave them intact (20). Our findings show that the policy had disproportionate impacts for people without versus people with schizophrenia, which can inform current decisions on HIV policies targeting vulnerable populations. For example, virtually no one in our study received HIV testing in a community mental health clinic, indicating a potential opportunity to develop interventions in mental health settings. Our findings can also contribute to future reviews of CDC recommendations. Second, in 2016, the Department of Health and Human Services (HHS) issued a joint informational bulletin aimed at improving HIV services for Medicaid beneficiaries (21), but people with mental illness were not included in the list of populations that could benefit from increased testing. Our report could inform HHS guidance on HIV testing among persons with mental illness. Third, the COVID-19 pandemic disrupted HIV services and underscored the existence of known weaknesses in public health infrastructure. This research provides key insight into the needs of a vulnerable population, which can inform policy makers, providers, and advocates who seek to strengthen public health care delivery. Fourth, the data represent the largest data set to examine HIV testing for people with schizophrenia within a representative cohort—two-thirds (67%) of U.S. adults with schizophrenia report having Medicaid (14). Given significant delays in access to Medicaid data, utilizing older data is common. Fifth, differences in the basic individual-level demographic characteristics we identified among those with schizophrenia likely have not markedly changed since 2012. For example, we noted similar female-to-male ratios of HIV testing rates in 2002 (1.54) and 2012 (1.60) among Medicaid beneficiaries with schizophrenia (Table 2). Finally, this work is critical given the limited research in this area.
Other study limitations included those common to observational and claims-based research (22). MAX data exclude billing data from behavioral health carveouts for multiple states, which may have resulted in missing data and therefore influenced our findings. In addition, MAX data do not include some laboratory billing data from managed care plans, although we had no reason to believe that this exclusion had differential effects on the two study cohorts and MAX data completeness increased during the study period (23). Although frequency matching accounted for demographic differences, unmeasured confounding was likely. Our cohort was restricted to Medicaid-only enrollees and did not represent dually eligible or commercially insured populations. Although Medicaid enrollees who were dually eligible for Medicare were excluded after matching, the similar distribution of matching factors between the cohorts suggested that selection bias was minimal. Diagnoses were assigned by clinicians, rather than with established instruments, and claims did not capture medical events that were not financed by Medicaid (e.g., HIV testing in correctional settings). Nevertheless, comparisons of Medicaid claims with state HIV and AIDS registry data have found that Medicaid claims have good sensitivity across subgroups, correctly identifying people whose HIV status is documented in the registry (24). Finally, the 2002–2012 data reported here did not allow for direct observation of contemporary HIV testing practices but enabled an examination of national growth patterns in HIV testing during a decade of advocacy efforts and implemented governmental policy changes intended to promote optimal testing. Thus, our findings provide an opportunity to look for patterns that are not evident in shorter time frames or more restricted groups and provide subgroup analyses that are not possible with more restricted cohorts.
Conclusions
This study reports HIV testing patterns and trends in a large national cohort of Medicaid-only enrollees with or without a diagnosis of schizophrenia. Despite long-standing efforts to raise awareness of high HIV prevalence among people with schizophrenia, HIV testing efforts fell short, with >90% of people with schizophrenia having no evidence of an HIV test in the most recent observation year. Medicaid and public mental health authorities could use incentives for integration of care to improve HIV testing among people with schizophrenia.
Future national HIV testing guidelines would benefit from referencing the higher rates of HIV and AIDS among individuals with schizophrenia, in addition to their higher-risk behaviors. Additionally, incorporating routine HIV testing into schizophrenia practice guidelines, alongside metabolic monitoring, could improve HIV outcomes for this population.
1. : Prevalence of HIV, hepatitis B, and hepatitis C in people with severe mental illness: a systematic review and meta-analysis. Lancet Psychiatry 2016; 3:40–48Crossref, Medline, Google Scholar
2. : 2012 UNAIDS Report on the Global AIDS Epidemic. Geneva, World Health Organization, 2012. https://www.unaids.org/en/resources/documents/2012/20121120_UNAIDS_Global_Report_2012. Accessed Jan 5, 2023 Google Scholar
3. : A multisite study of the prevalence of HIV with rapid testing in mental health settings. Am J Public Health 2014; 104:2377–2384Crossref, Medline, Google Scholar
4. : HIV risk behavior among adults with severe mental illness: a systematic review. Clin Psychol Rev 2005; 25:433–457Crossref, Medline, Google Scholar
5. : Substance abuse as a risk factor for HIV sexual risk behavior among persons with severe mental illness: review of evidence and exploration of mechanisms. Clin Psychol Sci Pract 2007; 14:23–33 Crossref, Google Scholar
6. : Primary and secondary HIV prevention among persons with severe mental illness: recent findings. Curr HIV/AIDS Rep 2015; 12:406–412Crossref, Medline, Google Scholar
7. : AIDS and People With Severe Mental Illness: A Handbook for Mental Health Professionals. New Haven, Yale University Press, 1996 Google Scholar
8. : Revised Recommendations for HIV Testing of Adults, Adolescents, and Pregnant Women in Health-Care Settings. Atlanta, Centers for Disease Control and Prevention, 2006. https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5514a1.htm. Accessed Jan 5, 2023 Google Scholar
9. : Community mental health’s role in the AIDS crisis. Community Ment Health J 1989; 25:185–196Crossref, Medline, Google Scholar
10. : Research on HIV, AIDS, and severe mental illness: recommendations from the NIMH National Conference. Clin Psychol Rev 1997; 17:327–331Crossref, Medline, Google Scholar
11. : HIV testing among individuals with a severe mental illness: review, suggestions for research, and clinical implications. Psychol Med 2009; 39:355–363Crossref, Medline, Google Scholar
12. : Increasing HIV testing in inpatient psychiatry. Psychosomatics 2018; 59:186–192Crossref, Medline, Google Scholar
13. : Low rates of HIV testing among adults with severe mental illness receiving care in community mental health settings. Psychiatr Serv 2017; 68:443–448Link, Google Scholar
14. : Health insurance coverage among persons with schizophrenia in the United States. Psychiatr Serv 2010; 61:830–834Link, Google Scholar
15. : Revised cost estimates of Medicaid recipients with serious mental illness and HIV-AIDS. Psychiatr Serv 2009; 60:974–977Link, Google Scholar
16. : Use of Medicaid data to explore community characteristics associated with HIV prevalence among beneficiaries with schizophrenia. Public Health Rep 2011; 126:89–101Crossref, Medline, Google Scholar
17. : Accuracy of diagnoses of schizophrenia in Medicaid claims. Hosp Community Psychiatry 1992; 43:69–71Abstract, Google Scholar
18. : Access to medical care among persons with psychotic and major affective disorders. Psychiatr Serv 2008; 59:847–852Link, Google Scholar
19. : Written informed-consent statutes and HIV testing. Am J Prev Med 2009; 37:57–63Crossref, Medline, Google Scholar
20. : Recommendations for HIV screening of gay, bisexual, and other men who have sex with men—United States, 2017. MMWR Morb Mortal Wkly Rep 2017; 66:830–832Crossref, Medline, Google Scholar
21. Opportunities to Improve HIV Prevention and Care Delivery to Medicaid and CHIP Beneficiaries. Washington, DC, Department of Health and Human Services, Centers for Medicare and Medicaid Services, Health Resources and Services Administration, Centers for Disease Control and Prevention, 2016. https://www.medicaid.gov/federal-policy-guidance/downloads/cib120116.pdfGoogle Scholar
22. : Studying prescription drug use and outcomes with Medicaid claims data: strengths, limitations, and strategies. Med Care 2007; 45:S58–S65Crossref, Medline, Google Scholar
23. : Internal validation of Medicaid Analytic eXtract (MAX) data capture for comprehensive managed care plan enrollees from 2007 to 2010. Pharmacoepidemiol Drug Saf 2018; 27:1067–1076Crossref, Medline, Google Scholar
24. : Sensitivity of an AIDS case-finding algorithm: who are we missing? Med Care 2004; 42:756–763Crossref, Medline, Google Scholar



