Systematic Quality Improvement and Metabolic Monitoring for Individuals Taking Antipsychotic Drugs
Abstract
Objective:
The authors sought to increase the rate of cardiometabolic monitoring for patients receiving antipsychotic drugs in an academic outpatient psychiatric clinic serving people with serious mental illness.
Methods:
Using a prospective quasi-experimental, interrupted time-series design with data from the electronic health record (EHR), the authors determined metabolic monitoring rates before, during, and after implementation of prespecified quality improvement (QI) measures between August 2016 and July 2017. QI measures included a combination of provider, patient, and staff education; systematic barrier reduction; and an EHR-based reminder system.
Results:
After 1 year of QI implementation, the rate of metabolic monitoring had increased from 33% to 49% (p<0.01) for the primary outcome measure (hemoglobin A1C and lipid panel). This increased monitoring rate was sustained for 27 months beyond the end of the QI intervention. More than 75% of providers did not find the QI reminders burdensome.
Conclusions:
Significant improvement in the rate of metabolic monitoring for people taking antipsychotic drugs can be achieved with little added burden on providers. Future research needs to assess the full range of patient, provider, and system barriers that prevent cardiometabolic monitoring for all individuals receiving antipsychotic drugs.
HIGHLIGHTS
People with serious mental illness taking antipsychotic drugs have high rates of cardiovascular and related metabolic disorders.
The authors describe the implementation of a 12-month quality improvement (QI) intervention aimed at improving cardiometabolic laboratory monitoring among people taking antipsychotic drugs in a large outpatient psychiatry clinic.
After 1 year of QI implementation, the rate of metabolic monitoring (hemoglobin A1C and lipid panel) had increased from 33% at baseline to 49% postintervention, and this increase was sustained 27 months after termination of active QI cycles.
The QI interventions were easy to implement and not viewed as burdensome by most providers.
Approximately 5.5% of the world’s population has serious mental illness (1), and individuals with serious mental illness die prematurely, with all-cause mortality 2.2-fold higher than that of individuals in the general population (2). Evidence suggests that two-thirds of these deaths occur from natural causes (2), and excess cardiovascular disorders represent the largest single contributor (3, 4). Moreover, the gap in life expectancy between those living with serious mental illness and those who are not appears to be increasing over time (5, 6). Despite significantly elevated rates of obesity, diabetes, hypertension, hyperlipidemia, and cardiovascular disorders (7, 8), many people with serious mental illness remain untreated for these conditions (9). Much evidence indicates that the general medical health needs of individuals with serious mental illness go unmet because of various individual-, provider-, and system-level factors (10–12).
There is particular concern about the role of antipsychotic drugs in accelerating the development of chronic general medical health problems such as obesity, hypertension, and elevated blood glucose among people with serious mental illness, given evidence that all antipsychotic drugs have the potential to cause weight gain and that all age groups are vulnerable (13–15). Accordingly, numerous guidelines for metabolic monitoring for people taking antipsychotic drugs have been developed, both in the United States and internationally; most guidelines focus on key risk factor assessments for cardiovascular disorders such as weight, blood pressure, and one or more measures of glucose and lipid metabolisms (8, 16, 17). Despite wide dissemination of these guidelines, available data indicate that the implementation of cardiometabolic monitoring remains inadequate (18, 19).
Few studies have addressed quality improvement (QI) strategies to improve metabolic laboratory monitoring in outpatient psychiatric clinics in the United States, with evidence of success limited primarily to improved documentation of monitoring (20–23). Key barriers to systematic metabolic monitoring, including electronic health record (EHR)-related dissatisfaction, have become significant concerns for providers (24–26). To address this gap in the literature, we conducted a QI project over 12 months to identify and address barriers to metabolic monitoring among individuals with serious mental illness taking antipsychotic drugs in an outpatient psychiatry clinic.
Methods
Design and Setting
To measure the impact of the QI intervention, we used a prospective, quasi-experimental interrupted time-series design to assess the rates of cardiometabolic monitoring for individuals prescribed antipsychotic drugs at an outpatient psychiatry clinic within an academic medical center.
In the process of establishing local guidelines, we found that height, weight, and blood pressure were monitored at the intervention clinic at >99.5% of all visits because these assessments had been incorporated into standard patient care before project implementation. Therefore, we designed our project to focus on improving guideline-based metabolic laboratory monitoring for persons taking antipsychotic drugs by targeting identified barriers to monitoring. The interventions included a combination of provider, patient, and staff education; systematic barrier reduction; and an EHR-based reminder system.
Monthly rates of metabolic monitoring were observed before and after the QI implementation between August 2016 and July 2017. The intervention clinic served many patients who were diagnosed as having a serious mental illness and who were receiving antipsychotic drugs. The clinic accepted any patient, regardless of insurance coverage. Phlebotomy services were located near the clinic within the medical center. Before QI implementation, the clinic did not perform systematic monitoring of cardiometabolic indexes for patients who were prescribed antipsychotic drugs. Because this study was part of a QI project, the Biomedical Institutional Review Board of the University of North Carolina at Chapel Hill determined that the study was exempt from review.
Interventions
The interventions were executed under the direction of the QI team, consisting of multiple stakeholders, including those in the fields of psychiatry (faculty, residents, and medical students), internal medicine (faculty), and biostatistics, as well as a clinic patient and family member representative.
Local Consensus Guideline
The current consensus guideline for antipsychotic-drug–associated cardiometabolic monitoring officially endorsed by the American Psychiatric Association, the American Association of Clinical Endocrinologists, and the American Diabetes Association was published in 2004 (17). This and other available guidelines were summarized and reconciled with current clinical practice in primary care settings for patients at high risk for developing cardiometabolic disorders in a series of multidisciplinary conferences (in the fields of psychiatry, internal medicine, family medicine, and endocrinology). The consensus was that medication providers in the intervention clinic (psychiatrists, nurse practitioners, and physician assistants) would screen for cardiometabolic adverse effects on an annual basis for patients receiving antipsychotic drugs and treat or refer them for follow-up with primary care providers for identified metabolic abnormalities. These guidelines were presented for further feedback from all providers in the clinic’s department of psychiatry and established after consensus was achieved.
The QI team provided education regarding the rationale for monitoring, the local guideline, and baseline monthly monitoring rates at departmental grand rounds, didactic sessions, and clinical staff meetings. Possible ambiguities were addressed over several feedback cycles. The finalized guideline was displayed prominently as posters in provider workspaces and in vital signs stations to encourage patient engagement with the QI project.
Patient- and Provider-Identified Barriers to Metabolic Monitoring
Baseline provider knowledge and attitudes related to monitoring of the adverse metabolic effects of antipsychotic drugs were assessed through a preintervention survey distributed to all department of psychiatry providers. Providers were encouraged to respond within 3 weeks. Providers were asked to identify barriers to obtaining antipsychotic drug–related metabolic monitoring laboratory tests.
A similar preintervention survey was distributed to all patients receiving antipsychotic medications at the intervention clinic over a 2-week period to identify patient-perceived barriers to laboratory monitoring. Results from both surveys were entered into Qualtrics software for analysis. Interventions were formulated and implemented on the basis of information obtained from these surveys.
Reminders for Metabolic Monitoring
Providers received a reminder before all patient visits about individuals who were due for metabolic laboratory monitoring. The reminder was initially provided on paper at the beginning of each clinic visit and then later integrated into the EHR system of Epic System Corporation. In brief, the list of patients who were due for cardiometabolic monitoring was generated by an algorithm that used clinical and administrative data from a central health care system repository. Each Thursday, a search was run on the data from all patients with an appointment scheduled for the following week at the intervention clinic. The EHRs of these patients were queried for whether they were receiving any antipsychotic drug, by using a live-updating list that cataloged all known antipsychotic drugs. Positive hits were then queried for the existence of appropriate metabolic monitoring, as described above, at different intervals to capture those with appropriate monitoring. After iterative search generation that included adjusting the intervals to search, reminder notes were provided for patients who had no record of EHR-based metabolic monitoring in the past 15 months from the date of the scheduled appointment. A 15-month observation interval rather than a 12-month interval was chosen to avoid overidentifying patients as nonadherent with testing as long as their annual testing was performed in close proximity to 12 months. The EHR reminder was passive, visible to all members of the clinic regardless of their role, and was placed in a portion of the EHR that would be visible without access to the patients’ medical records.
Other interventions to target key measures included a flier with directions from the intervention clinic to the phlebotomy laboratory distributed to patients by clinic staff and a precompleted release-of-information request form specific to laboratory monitoring with a subsequent clinic process to obtain results performed at outside facilities. The form was distributed to all patients who indicated that an outside provider was performing metabolic monitoring. A process with a designated staff member to follow up on these forms was created.
Measures
The primary outcome measure was the proportion of patients visiting the clinic each month who had obtained appropriate metabolic monitoring. Guideline-concordant cardiometabolic laboratory monitoring was defined as having both hemoglobin A1C (HbA1C), or fasting blood glucose, and lipid panel (fasting or nonfasting) performed within 15 months of any patient visit.
Data Analysis
Given the dynamic nature of any clinic population, the proportion of patients with guideline-concordant metabolic monitoring at any given time is susceptible to change for reasons unrelated to a QI intervention. To increase the likelihood that any changes observed were due to the intervention, we determined the proportions of patients with appropriate monitoring per month for 6 months before the QI intervention to ascertain a stable baseline, as well as monthly during the intervention. We determined the change in the ongoing monitoring rate before the intervention. We hypothesized that implementation of the QI project would lead to an increase in the rate of metabolic monitoring.
For testing the primary hypothesis, generalized linear mixed-effects models were fit to predict the proportion of patients monitored each month. Because many patients were monitored more than once in the data set, a random intercept for each patient was included to account for nonindependence of observations. The distribution of our dependent variable was binomial (guideline concordant vs. nonconcordant), and a logit link was used. We further controlled for patient-level covariates, including demographic characteristics (i.e., age, sex, race, ethnicity, and marital status), prescribed antipsychotic drugs, and diagnoses. The model was fit to three monitoring outcomes: HbA1C, lipids, and both HbA1C and lipids (primary outcome). These models afforded interpretations that applied to the probability of metabolic monitoring in individual patients, given their specific characteristics. We conducted statistical analyses with StataSE 16 (StataCorp). The significance levels took into account the repeated measures resulting from such analyses.
A secondary hypothesis was whether any improvement in monitoring rates would be sustained beyond the QI project duration. For this analysis, the mean monitoring rates over two 6-month periods were compared, one immediately at the conclusion of the QI project (August 2017–January 2018) and another 21 months later (May 2019–October 2019).
Results
Demographic characteristics of 1,719 patients who received at least one antipsychotic drug prescription during the 12-month interval of the QI project are provided in Table 1. Diagnostic information represented clinical diagnoses extracted from the EHR. Many patients had overlapping psychiatric diagnoses, in particular, diagnoses of mood and anxiety disorders; 30% of the patients (N=517) had a schizophrenia spectrum disorder. Notably, many people in the clinic without a primary psychotic disorder received antipsychotic drugs.
| Characteristic | N | % |
|---|---|---|
| Age (M±SD) | 44.4±19.2 | |
| Gender | ||
| Female | 1,019 | 59 |
| Male | 700 | 41 |
| Partner status | ||
| Single | 872 | 51 |
| Partnered | 523 | 30 |
| Divorced, separated, or widowed | 240 | 14 |
| Race | ||
| Caucasian | 1,240 | 72 |
| Black or African American | 267 | 16 |
| Other | 212 | 12 |
| Ethnicity | ||
| Not Hispanic | 1,548 | 90 |
| Hispanic | 65 | 4 |
| Unknown | 106 | 6 |
| Psychiatric diagnosisa | ||
| Anxiety disorder | 1,199 | 70 |
| Major depressive disorder | 1,161 | 68 |
| Bipolar disorder | 575 | 33 |
| Other mood disorder | 492 | 29 |
| Psychotic disorder | 571 | 33 |
| Obsessive-compulsive disorder or tic disorder | 189 | 11 |
| Neurocognitive disorder | 135 | 8 |
| Neurodevelopmental disorder | 117 | 7 |
| Conduct disorder | 109 | 6 |
| Eating disorder | 97 | 6 |
| Medical cause | 299 | 17 |
| Cardiometabolic diagnosisa | ||
| Hypertension | 678 | 40 |
| Dyslipidemia | 506 | 29 |
| Hyperglycemia | 207 | 12 |
| Diabetes | 285 | 17 |
| Cardiovascular disease | 201 | 12 |
| Cerebrovascular disease | 180 | 11 |
TABLE 1. Demographic characteristics for 1,719 patients with serious mental illness prescribed antipsychotic drugs during the quality improvement period
Metabolic Laboratory Monitoring Rates
Pareto charts based on the responses to the surveys on patient- and provider-perceived barriers to cardiometabolic monitoring are shown in Figure 1. Patients noted as the biggest monitoring barrier the fact that the monitoring was not recommended by their provider, and providers reported that some patients did not want to undergo laboratory testing.
During the 6-month preintervention period, the percentage of patients taking antipsychotic drugs who received guideline-concordant metabolic monitoring ranged from 33% to 38%. The clinic had no statistically significant change in metabolic monitoring rates during this 6-month baseline period preceding the initiation of the QI intervention. During the intervention period, monitoring rates significantly increased for all three outcomes compared with baseline (HbA1C: b=0.16, z=8.9, p<0.001; lipids: b=0.12, z=7.6, p<0.001; HbA1C and lipids [primary outcome]: b=0.15, z=9.1, p<0.001). Consistent with these results, the data in Figure 2, depicting monitoring over time, indicated a shift above the respective baseline median lines for each of the three outcome variables. Guideline-concordant monitoring of the combined measures of HbA1C and lipids increased from 33% at the beginning of the QI intervention period (August 2016) to 49% at the end of the intervention (July 2017).
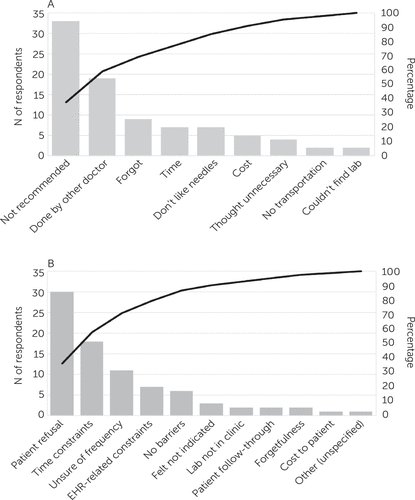
FIGURE 1. Beliefs of 64 patients and 45 providers regarding barriers to metabolic laboratory monitoringa
aPatient-perceived (A) and provider-perceived (B) barriers to laboratory monitoring. The Pareto charts are based on surveys distributed to 294 patients prescribed antipsychotics, of whom 64 responded (A), and to 67 providers prescribing antipsychotics at the intervention clinic, of whom 45 responded (B). Both surveys allowed for multiple selections. EHR, electronic health record.
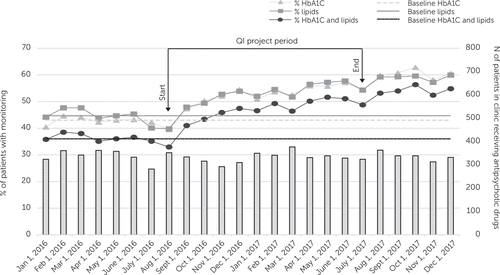
FIGURE 2. Metabolic laboratory monitoring before, during, and after implementation of the quality improvement (QI) interventiona
aThe three solid lines represent rates of monitoring for hemoglobin A1C (HbA1C), lipids, and both HbA1C and lipids as a percentage of the patients receiving antipsychotic drugs per month. The three dashed lines represent the median monitoring rates for HbA1C, lipids, and both during the baseline period. Bars represent the number of individual patients with appointments per month who received antipsychotic drugs.
To address the possibility that the change in monitoring rates could have been due to factors unrelated to the QI project, we identified a comparison clinic within the same medical system at a different geographic location that also served a large population of patients with serious mental illness receiving antipsychotic drugs, previously described by Perrin et al. (27). The aforementioned model was applied to both the intervention clinic and the comparison clinic and analyzed as a three-way interaction among time, clinic, and QI project initiation.
At the comparison clinic, baseline rates of metabolic monitoring were lower than at the intervention clinic, ranging from 17% to 23% during the 6-month preintervention period. Furthermore, the rate of monitoring slowly increased, unlike the rate at the intervention clinic, which had a flat pre-QI baseline. Monitoring rates at the comparison clinic continued to slowly increase at approximately the same rate during the QI phase of the intervention clinic and showed no acceleration or deceleration. The analysis of the three-way interaction between clinic, time, and intervention was significant in all three models (HbA1C: b=−0.13, z=−4.4, p<0.001; lipid: b=−0.06, z=−2.3, p=0.02; HbA1C and lipids: b=−0.12, z=−4.2, p<0.001), indicating a significant change in the monitoring rate at the intervention clinic that differed from the more steady change in the rate at the comparison clinic during the QI application phase. The patient population receiving antipsychotic drugs at the two clinics remained stable over the study period.
To address the possibility that the increase in monitoring rates could be accounted for by measurement bias, we conducted chart reviews. All patients who were prescribed any antipsychotic drugs and who did not have annual HbA1C or lipid monitoring were identified. Only two instances of false positives were noted, and these were due to laboratory tests having been performed after reminders had been entered into the EHR.
Attempts at obtaining laboratory results from outside laboratories and nonaffiliated clinics proved challenging. For the first 50 forms completed, sent, and followed up with multiple phone calls, only nine laboratories and clinics returned guideline-concordant results. Given the low rate of return relative to the effort required, this clerical process was eventually discontinued. The forms remained available for use by individual providers. Providers were also instructed to provide feedback to the QI team for inappropriate monitoring reminders (i.e., EHR reminders despite evidence of completed laboratory testing). Only four such reports were received, and these were also due to laboratory tests having been performed after reminders had been entered.
The providers’ perception of burden due to the QI efforts were assessed in a postintervention survey (Table 2). Most providers (34 of 44, 77%) did not find the reminders to be burdensome. To determine whether the improvements observed during the QI initiative were sustained, mean rates of metabolic monitoring from the 6 months immediately after the active QI phase (August 2017–January 2018) were compared with the mean rates of monitoring 22–27 months after the active QI phase (May 2019–October 2019). The monitoring rates were sustained for HbA1C (60.2% vs. 60.7%, respectively), lipids (58.9% vs. 60.3%, respectively), and both HbA1C and lipids (54.0% vs. 54.2%, respectively).
| Response | N | % |
|---|---|---|
| Strongly disagree | 8 | 18 |
| Disagree | 9 | 20 |
| Somewhat disagree | 4 | 9 |
| Neither agree nor disagree | 14 | 31 |
| Somewhat agree | 7 | 16 |
| Agree | 2 | 4 |
| Strongly agree | 1 | 2 |
TABLE 2. Responses of 45 providers to a survey on perception of provider burden of the quality improvement (QI) projecta
Discussion and Conclusions
Individuals who have serious mental illness often die prematurely, and evidence suggests that excess cardiovascular disease represents the single largest contributor to this increased mortality rate (3). Undiagnosed cardiometabolic disorders (e.g., hyperlipidemia, diabetes, and hypertension) among people taking antipsychotic medications likely contribute to reduced life spans in this population. The findings of this study indicate that the QI intervention, consisting of education and EHR-based reminders, increased cardiometabolic monitoring rates in an outpatient psychiatric clinic serving patients with serious mental illness who take antipsychotic drugs. The intervention was associated with increased monitoring of the combined measures of HbA1C and lipids from 33% to 49% of all patients receiving antipsychotic drugs within the previous 12-month period. These results provide evidence that psychiatric providers can and should have an important frontline role in identifying cardiometabolic abnormalities among people with serious mental illness receiving antipsychotic drugs. We are not aware of similar comparison studies performed in outpatient settings in the United States. Most other studies indicating the effectiveness of reminder- and education-based interventions took place in countries with substantially different health care systems (28–31) or were limited to inpatient settings (32–34).
Advantages of our EHR-based reminder system included the ease with which it was disseminated, as well as how it functioned as a reminder: there was no hard stop, and it was not presented as a warning by the system. The finding that our approach to reminders did not result in a perception of burden among providers stands in contrast to other studies in which having too many EHR-based reminders was associated with provider fatigue (35) and burnout (25). The reminders were the product of iterative cycles of change, including format (from paper to electronic), location (from one physician workroom to the EHR), and length (limited to 16 characters). The final EHR reminder required 15–30 minutes per week of clerical staff time. It is encouraging to note that the QI initiative was sustainable and required limited effort relative to its benefits. We believe that the wall-mounted poster guidelines and the EHR-based reminders for metabolic monitoring represented key components for this sustainability.
Numerous identified barriers that we attempted to solve locally would have benefited from larger systematic changes. We could obtain guideline-concordant results only for nine of 50 patients who reported having an outside provider for monitoring. A centralized laboratory reporting system comparable to the Risk Evaluation and Mitigation Strategy system of the U.S. Food and Drug Administration would represent a possible approach to reduce the burden of follow-up from individual clinics, reduce redundant blood draws, and maximize adherence to monitoring guidelines to improve health for people taking antipsychotic drugs. Furthermore, the number of providers who manually entered outside lab results into the EHR remained low throughout the project. Further engagement with relevant stakeholders and partners in the broader health care system and community to promote cardiometabolic monitoring as part of community-based lifestyle intervention efforts and to encourage information sharing across these systems would likely also improve outcomes.
Many factors can affect the ability of individual clinics to effectively implement metabolic monitoring programs for patients taking antipsychotic drugs. For example, some clinics do not have the staff or the necessary equipment to routinely obtain vital signs (21, 23). As most hospital-based outpatient psychiatric clinics, our intervention clinic has limited resources, such as a lack of in-clinic phlebotomy services, time constraints on both patients and staff, diverse demographic patient characteristics, and varied reimbursement forms.
Finally, although this QI intervention successfully improved rates of metabolic monitoring, further improvements can likely be gained through additional reductions in monitoring barriers. Despite the improvements seen with implementation of this QI project, up to 45% of patients in the intervention clinic did not have guideline-concordant monitoring. Potential approaches for further improvements include integrated primary care providers within psychiatric clinics, an approach with strong evidence for improving metabolic monitoring, as well as other primary care outcome measures (36, 37). We recognize that monitoring alone is insufficient but that cardiometabolic monitoring during antipsychotic drug treatment is essential if meaningful improvements are to be achieved in overall health and reduced mortality rate for this vulnerable and often difficult-to-engage population. In addition to monitoring, initiating appropriate interventions when indicated, ensuring that all patients have a primary care provider, and referring patients to specialists when metabolic abnormalities are detected represent key components of comprehensive health care for people with serious mental illness who are taking antipsychotic drugs.
1 : The global burden of mental, neurological and substance use disorders: an analysis from the Global Burden of Disease Study 2010 . PLoS One 2015 ; 10 : e0116820 Crossref, Medline, Google Scholar
2 : Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis . JAMA Psychiatry 2015 ; 72 : 334 – 341 Crossref, Medline, Google Scholar
3 : Prevalence, incidence and mortality from cardiovascular disease in patients with pooled and specific severe mental illness: a large-scale meta-analysis of 3,211,768 patients and 113,383,368 controls . World Psychiatry 2017 ; 16 : 163 – 180 Crossref, Medline, Google Scholar
4 : Premature mortality among adults with schizophrenia in the United States . JAMA Psychiatry 2015 ; 72 : 1172 – 1181 Crossref, Medline, Google Scholar
5 : The gap in life expectancy from preventable physical illness in psychiatric patients in Western Australia: retrospective analysis of population based registers . BMJ 2013 ; 346 : f2539 Crossref, Medline, Google Scholar
6 : A systematic review of mortality in schizophrenia: is the differential mortality gap worsening over time? Arch Gen Psychiatry 2007 ; 64 : 1123 – 1131 Crossref, Medline, Google Scholar
7 : Metabolic syndrome in people with schizophrenia: a review . World Psychiatry 2009 ; 8 : 15 – 22 Crossref, Medline, Google Scholar
8 : Guidelines for screening and monitoring of cardiometabolic risk in schizophrenia: systematic evaluation . Br J Psychiatry 2011 ; 199 : 99 – 105 Crossref, Medline, Google Scholar
9 : Low rates of treatment for hypertension, dyslipidemia and diabetes in schizophrenia: data from the CATIE schizophrenia trial sample at baseline . Schizophr Res 2006 ; 86 : 15 – 22 Crossref, Medline, Google Scholar
10 : Congruencies in increased mortality rates, years of potential life lost, and causes of death among public mental health clients in eight states . Prev Chronic Dis 2006 ; 3 : A42 Medline, Google Scholar
11 : Quality of preventive medical care for patients with mental disorders . Med Care 2002 ; 40 : 129 – 136 Crossref, Medline, Google Scholar
12 : Use of general medical services among Medicaid patients with severe and persistent mental illness . Psychiatr Serv 2005 ; 56 : 458 – 462 Link, Google Scholar
13 : Antipsychotic-induced weight gain: a comprehensive research synthesis . Am J Psychiatry 1999 ; 156 : 1686 – 1696 Link, Google Scholar
14 : Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: a systematic review and meta-analysis . Schizophr Res 2010 ; 123 : 225 – 233 Crossref, Medline, Google Scholar
15 : Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents . JAMA 2009 ; 302 : 1765 – 1773 Crossref, Medline, Google Scholar
16 : Physical health monitoring of patients with schizophrenia . Am J Psychiatry 2004 ; 161 : 1334 – 1349 Link, Google Scholar
17 Consensus development conference on antipsychotic drugs and obesity and diabetes . Diabetes Care 2004 ; 27 : 596 – 601 Crossref, Medline, Google Scholar
18 : Monitoring of recommended metabolic laboratory parameters among Medicaid recipients on second-generation antipsychotics in federally qualified health centers . Ann Pharmacother 2018 ; 52 : 160 – 165 Crossref, Medline, Google Scholar
19 : Measurement of metabolic monitoring in youth and adult Medicaid recipients prescribed antipsychotics . J Manag Care Spec Pharm 2015 ; 21 : 769 – 777 Crossref, Medline, Google Scholar
20 : A randomized controlled trial of a patient-centered approach to improve screening for the metabolic side effects of antipsychotic medications . Community Ment Health J 2017 ; 53 : 163 – 175 Crossref, Medline, Google Scholar
21 : Quality improvement in resident education: a pilot project to mitigate metabolic side effects from atypical antipsychotic medications in youth . BMJ Qual Improv Rep 2015 ; 4 : u208804.w3544 Crossref, Medline, Google Scholar
22 : Antipsychotic cardiometabolic side effect monitoring in a state community mental health system . Community Ment Health J 2015 ; 51 : 685 – 694 Crossref, Medline, Google Scholar
23 : Impact of a metabolic screening bundle on rates of screening for metabolic syndrome in a psychiatry resident outpatient clinic . Acad Psychiatry 2012 ; 36 : 118 – 121 Crossref, Medline, Google Scholar
24 : More screen time, less face time—implications for EHR design . J Eval Clin Pract 2014 ; 20 : 896 – 901 Crossref, Medline, Google Scholar
25 : Electronic health record alert-related workload as a predictor of burnout in primary care providers . Appl Clin Inform 2017 ; 8 : 686 – 697 Crossref, Medline, Google Scholar
26 What’s ruining medicine for physicians: difficulty using EHRs . Med Econ 2018 ; 95 https://www.medicaleconomics.com/view/whats-ruining-medicine-physicians-difficulty-using-ehrs Google Scholar
27 : A model of enhanced primary care for patients with severe mental illness . N C Med J 2018 ; 79 : 240 – 244 Medline, Google Scholar
28 : Using an integrated care pathway for late-life schizophrenia improves monitoring of adverse effects of antipsychotics and reduces antipsychotic polypharmacy . Am J Geriatr Psychiatry 2019 ; 27 : 84 – 90 Crossref, Medline, Google Scholar
29 : Monitoring and management of metabolic abnormalities: mixed-method evaluation of a successful intervention . Australas Psychiatry 2014 ; 22 : 248 – 253 Crossref, Medline, Google Scholar
30 : Targeted intervention to improve monitoring of antipsychotic-induced weight gain and metabolic disturbance in first episode psychosis . Aust N Z J Psychiatry 2011 ; 45 : 740 – 748 Crossref, Medline, Google Scholar
31 : Metabolic monitoring training program implementation in the community setting was associated with improved monitoring in second-generation antipsychotic-treated children . Can J Psychiatry 2012 ; 57 : 292 – 299 Crossref, Medline, Google Scholar
32 : Persistence of metabolic monitoring for psychiatry inpatients treated with second-generation antipsychotics utilizing a computer-based intervention . J Clin Pharm Ther 2016 ; 41 : 209 – 213 Crossref, Medline, Google Scholar
33 : Is metabolic syndrome on the radar? Improving real-time detection of metabolic syndrome and physician response by computerized scan of the electronic medical record . Prim Care Companion CNS Disord 2016 ; 18 :
34 : Monitoring of physical health parameters for inpatients on a child and adolescent mental health unit receiving regular antipsychotic therapy . BMJ Qual Improv Rep 2015 ; 4 : u202645.w3700 Crossref, Medline, Google Scholar
35 : Alert fatigue in electronic health records . JAAPA 2015 ; 28 : 41 – 42 Crossref, Medline, Google Scholar
36 : A randomized trial of medical care management for community mental health settings: the Primary Care Access, Referral, and Evaluation (PCARE) study . Am J Psychiatry 2010 ; 167 : 151 – 159 Link, Google Scholar
37 : Randomized trial of an integrated behavioral health home: the Health Outcomes Management and Evaluation (HOME) Study . Am J Psychiatry 2017 ; 174 : 246 – 255 Link, Google Scholar



