Scale-Up Study Protocol of the Implementation of a Mobile Health SBIRT Approach for Alcohol Use Reduction in Mozambique
Abstract
Background:
Hazardous drinking imposes a major public health burden worldwide, especially in low-income countries such as Mozambique. Implementation of the Screening, Brief Intervention, Referral to Treatment (SBIRT) approach to address problem drinking is recommended. However, evidence regarding the best strategies to implement SBIRT at scale is needed.
Methods:
Guided by the Reach Effectiveness Adoption Implementation Maintenance model, the authors will conduct a 2-year, cluster-randomized, hybrid type-2 implementation-effectiveness trial in 12 districts in Mozambique evaluating implementation, clinical effectiveness, outcomes, and cost. Eight districts will be randomly assigned to a mobile application–based health SBIRT condition and four to SBIRT–Conventional Training and Supervision. Interventions will be delivered by clinic-based community health workers. The Consolidated Framework for Implementation Research will guide the authors’ mixed-methods assessments throughout the study.
Results:
The study arm showing better cost-effectiveness will be scaled up in the other arms’ districts. During this 12-month scale-up phase, Ministry of Health personnel will be charged with providing training, clinical activities, and supervision in all 12 districts without research team support. The SBIRT scale-up phase is critical to identify facilitators and barriers for tracking internal and external factors in clinics that continue using the superior arm and those that switch to it.
Next steps:
In a multistep process with stakeholders from multiple sectors, outcomes and lessons learned from this study will inform the development of an implementation tool kit to guide SBIRT scale-up of community services addressing hazardous drinking in other low- and middle-income countries and low-resource settings in high-income countries.
HIGHLIGHTS
To reduce hazardous drinking in Mozambique, this study compares the Screening, Brief Intervention, Referral to Treatment–Conventional Training and Supervision (SBIRT-CTS) approach with a mobile health version of SBIRT (mSBIRT), examining community-level implementation, clinical outcomes, and cost-effectiveness.
The use of mSBIRT is envisioned as a cost-effective, sustainable delivery mechanism for evidence-based services in low- and middle-income countries.
This study explores a causal model and predictors of SBIRT implementation success by combining psychological theories of behavior change with organizational theories.
Hazardous drinking, that is, alcohol consumption that places individuals at risk for adverse health events, is a major public health challenge, particularly in low- and middle-income countries (LMICs) (1–3). Alcohol misuse is avoidable, yet results in high morbidity and mortality rates and ranks as the third-leading risk factor for poor health globally (1–4). Harmful alcohol use contributes to >200 types of diseases and injuries. Alcohol’s impact is worst among people in poor populations and in LMICs where disease burden per liter of alcohol consumed is greater than among individuals in wealthy populations (2, 5, 6).
Editor’s Note: In partnership with Milton L. Wainberg, M.D., Psychiatric Services is publishing protocols to address the gap between global mental health research and treatment. These protocols present large-scale, global mental health implementation studies soon to begin or under way. Taking an implementation science approach, the protocols describe key design and analytic choices for delivery of evidence-based practices to improve global mental health care. This series represents the best of our current science, and we hope these articles inform and inspire.
Mozambique is a low-income country where half of the population lives below the poverty line as set by the World Bank and disease burden is high. Although research on alcohol use in Mozambique is scarce, recent studies have revealed that binge drinking (episodes of excessive drinking, i.e., four or more drinks per episode for women and five or more drinks per episode for men) is frequent among drinkers in Mozambique. Two recent studies with Mozambicans ages 25–64 years found that 24%−29% of women and 49%−58% of men were current drinkers (having consumed at least one drink in the past year) (7, 8). Approximately 60% of current drinkers consumed alcohol at least once or twice a week, and >40% reported binge drinking in the previous week. Three-quarters of drinkers reported consuming home-distilled traditional alcohol beverages with high alcohol content, which was strongly associated with binge drinking. Having depression and experiencing the death of a child were associated with hazardous drinking (a score of >4 on the Alcohol Use Disorders Identification Test [AUDIT]) (9).
Mozambique’s HIV prevalence has increased by 15% since 2009 (10). Alcohol is implicated in behaviors that increase the risk for contracting HIV and in poor HIV care adherence (11). HIV is the second largest category of alcohol-attributable disability-adjusted life years (DALYs); alcohol-attributable disease burden worsens if the impact of alcohol consumption on HIV incidence and course is considered, with alcohol being responsible for 6.4% of all deaths and 4.7% of all DALYs lost in the African region (12). Thus, decreasing hazardous drinking could improve HIV care outcomes.
The World Health Organization’s Mental Health Gap Action Programme (mhGAP) guidelines (13, 14) recommend using the Screening, Brief Intervention, and Referral to Treatment (SBIRT) (15) approach to reduce hazardous drinking. SBIRT is typically integrated into medical care to address substance use disorders (16). However, there are challenges to implementing SBIRT with fidelity, obtaining the effectiveness observed in trials, and sustaining SBIRT (17), particularly in LMICs (18). Hazardous drinking services (HDS) in Mozambique are delivered in specialty clinics by psychiatric technicians trained in SBIRT and other HDS informed by mhGAP evidence. With one specialty clinic per district, each with 50,000–150,000 inhabitants, most individuals who engage in hazardous drinking are not served. Our study aims to address this challenge through work with the Mozambican Ministry of Health to task-shift SBIRT to community health workers (CHWs). We will build on the Ministry of Health’s mhGAP-Epilepsy Program (19) and our community mental health trial, funded recently by the National Institute of Mental Health, which links study data to Mozambique’s electronic medical record system (e-saúde) to track the impact of two SBIRT approaches on comorbid conditions, including HIV and tuberculosis (TB).
Mobile health technologies (mHealth) (20), such as the mobile SBIRT (mSBIRT) application (21), are a promising tool for widespread, cost-effective, and sustainable health service delivery in LMICs (20, 22–24). The mSBIRT application was designed for use by health care providers. It assists providers in quickly assessing a patient’s alcohol use risk level and guides providers through a brief intervention that is tailored to the patient’s responses. For LMICs such as Mozambique, identifying the most appropriate strategy to implement SBIRT in community HDS is crucial. Capitalizing on Mozambique’s task-shifting strategies and commitment to inform HDS scale-up with local treatment guidelines (i.e., SBIRT and mhGAP), this study will scale up SBIRT in the community. We will compare mSBIRT with the SBIRT–Conventional Training and Supervision (SBIRT-CTS) strategy to determine the most cost-effective approach for expansion and for overcoming implementation barriers.
Community Implementation of SBIRT using Technology for Alcohol use Reduction in Mozambique (Community I-STAR Mozambique) comprises a 2-year, cluster-randomized, hybrid implementation-effectiveness type-2 trial in 12 districts evaluated by mixed-methods analyses (Table 1). Our first two aims will be accomplished throughout the trial. Aim 1 is to conduct an implementation impact evaluation by using the Reach, Effectiveness, Adoption, Implementation, Maintenance (RE-AIM) model and compare the adapted mSBIRT with SBIRT-CTS in terms of reach (primary outcome), adoption, implementation fidelity, and maintenance over time. Aim 2 is to compare clinical effectiveness and cost-effectiveness of mSBIRT and SBIRT-CTS overall and by patient’s gender, age, and urbanicity (25). Aim 3 will be accomplished throughout the trial and the subsequent scale-up phase (i.e., when the most effective strategy is expanded to other districts) and will identify organizational and clinician-level factors that affect SBIRT implementation and effectiveness. Design solutions that are responsive to key challenges and advantages of the study are detailed in Box 1.
| Year 1 | Year 2 | Year 3 | Year 4 | Year 5 | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Milestone | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 |
| Start | ✔ | |||||||||||||||||||
| Personnel training, IRB approvals, measure finalizationa | ✔ | |||||||||||||||||||
| Finalizing SBIRT-CTS and mSBIRT training and supervision manuals and pilotingb | ✔ | ✔ | ✔ | |||||||||||||||||
| Finalizing data collection and management and manual of procedures | ✔ | |||||||||||||||||||
| Training of trainers and harmonizing training sessions | ✔ | |||||||||||||||||||
| Effectiveness-implementation type-2 RCT (aim 1 and 2)c | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ||||||||||||
| RCT implementation phasec | ✔ | ✔ | ✔ | ✔ | ||||||||||||||||
| Training of CHWs and PCPsd | ✔ | ✔ | ||||||||||||||||||
| Performing services, consultations, monitoring, feedback | ✔ | ✔ | ||||||||||||||||||
| RCT sustainability phasec | ✔ | ✔ | ✔ | ✔ | ||||||||||||||||
| Assessing competency | ✔ | ✔ | ✔ | ✔ | ||||||||||||||||
| Cost-effectiveness data analysis | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ||||||||||||||
| Scale-up of most effective arm and crossover | ✔ | ✔ | ✔ | ✔ | ||||||||||||||||
| Mixed-methods process evaluation (aim 3) | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | |||||||
| Conducting focus groups and interviews | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | |||||||
| Ongoing quantitative and qualitative data analyses | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | |||||||
| Finalizing tool kit development | ✔ | ✔ | ✔ | |||||||||||||||||
| Conducting policy workshop | ✔ | |||||||||||||||||||
TABLE 1. Time line and milestones of the Community Implementation of SBIRT using Technology for Alcohol use Reduction (I-STAR) in Mozambique trial, by yearly quarter
We will determine which study arm is superior according to implementation impact, effectiveness (clinical and cost), and implementation process. We chose the RE-AIM framework for aims 1 and 2 because it is widely studied and identifies five constructs that affect the quality, speed, and public health impact of efforts to translate research into practice: reach the intended target population; efficacy and effectiveness; adoption by target staff, settings, and institutions; implementation fidelity; and maintenance of intervention effects (26, 27). For a broader understanding of the implementation process (aim 3), we chose the Consolidated Framework for Implementation Research (CFIR) (28) to guide qualitative exploration of factors affecting implementation. CFIR synthesizes implementation science frameworks into five domains: intervention characteristics, outer setting (an organization’s economic, political, and social context), inner setting, characteristics of individuals involved in implementation, and implementation process.
BOX 1. Key challenges, advantages, and design solutions of Community Implementation of SBIRT using Technology for Alcohol use Reduction (I-STAR) in Mozambique
Challenges
Limited access to treatment services that address hazardous drinking and that are currently provided by psychiatric technicians at specialty clinics at the district level with only one specialty clinic per district.
Effectiveness, fidelity, and sustainability challenges in using Screening, Brief Intervention, Referral to Treatment (SBIRT) as part of hazardous drinking services in low- and middle-income countries (LMICs).
Untested comparative effectiveness and cost-effectiveness of mobile health technology and conventional training and supervision approaches.
Advantages
Mozambique’s Ministry of Health is involved in capacity building of implementation science research.
Task-shifting strategic policy is already in place, and psychiatric technicians, supervised by mental health specialists, provide mental health services in urban areas.
Mobile health SBIRT (mSBIRT) has been developed by research partners in the United States.
Design Solutions
Implement SBIRT for the prevention and management of hazardous drinking at the community level with delivery by nonspecialized providers in severely resource-limited settings.
Use mSBIRT as a cost-effective, sustainable delivery mechanism for evidence-based services in LMICs.
Use surrogate respondents as part of a public health intervention to identify household members who may be at risk for hazardous drinking.
Explore causal predictors of implementation success by combining psychological theories of behavior change with organizational theories.
Use state-of-the-art implementation measures to examine the implementation, effectiveness, and cost-effectiveness of two SBIRT delivery strategies during the implementation and sustainability phases, followed by evaluation of scale-up of the more cost-effective strategy.
However, neither RE-AIM nor CFIR posit causal links among variables. Specifically, they do not describe how organization- and provider-level factors interact to affect implementation. To address this limitation, we borrow from and expand upon the work of Williams and Glisson (Figure 1) (29), who combined organizational factors with psychological variables from the theory of planned behavior (25). We propose that the most proximal variable to CHWs’ and supervisors’ use of SBIRT is their intention to use it. Intentions are influenced by four determinants: attitudes (perceptions of SBIRT), descriptive norms (belief that people like me use SBIRT), injunctive norms (belief that important others expect me to use SBIRT), and self-efficacy (perception that providers can effectively perform SBIRT). We hypothesize that organizational factors influence the determinants of intentions and moderate the association between intentions and use of SBIRT. That is, even the strongest intentions may be thwarted if organizational resources do not support acting on those intentions. Our hypotheses are the following:
Hypothesis 1: RE-AIM Implementation Impact Evaluation
mSBIRT will yield significantly better RE-AIM implementation outcomes than will SBIRT-CTS. The primary outcome is reach (percentage of those in need who can access care). Secondary outcomes are adoption (percentage of clinic staff using SBIRT), implementation fidelity (adherence to model and strategy), and maintenance (use over 2 years) at months 12, 18, and 24.
Hypothesis 2: RE-AIM Clinical Effectiveness and Cost-Effectiveness Evaluation
First, clients exposed to mSBIRT will have greater reduction in hazardous drinking and improved health-related quality of life than those exposed to SBIRT-CTS at months 12, 18, and 24. Second, hazardous drinking reduction due to SBIRT will improve health-related outcomes, including HIV-related outcomes (e.g., reduced viral load) for people living with HIV at months 12, 18, and 24. Third, mSBIRT will be more cost-effective than SBIRT-CTS at months 12, 18, and 24.
Hypothesis 3: Implementation Process Evaluation
First, location of the organization (urban, suburban, or rural), culture, and climate will affect clinicians’ attitudes, perceived norms, and self-efficacy regarding SBIRT use, which will in turn affect intentions to use SBIRT. Second, clinicians’ demographic characteristics and intentions to use SBIRT will affect its adoption and use. Third, resources will moderate the association between clinicians’ intentions and implementation.
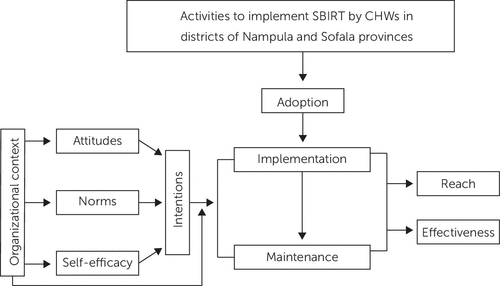
FIGURE 1. Applying organizational and psychological theories to implementation of the Screening, Brief Intervention, Referral to Treatment (SBIRT) approacha
aAdapted from Glasgow et al. (27) and Williams and Glisson (29). CHWs, community health workers.
Methods
Overview
We obtained ethical approval from institutional review boards at New York State Psychiatric Institute and the Mozambican Institute for Health Education and Research. We will conduct a 2-year, cluster-randomized, hybrid implementation-effectiveness type-2 trial (30) in 12 districts. CHWs will deliver the intervention in both intervention arms. A 2:1 randomization of districts will be used, with eight districts randomly assigned to mSBIRT and four to SBIRT-CTS. Randomization will be stratified on the basis of district size (i.e., number of CHWs) to ensure proper balance. The more effective strategy will be scaled up to the other arm in study year 3. Guided by the CFIR (28), we will use mixed-methods assessments throughout the study to identify the factors affecting implementation. This study is embedded in a larger study of a scale-up of comprehensive mental health care (31).
Study Sites
The study will be conducted in two Mozambican provinces—Nampula and Sofala. Nampula province is located in northern Mozambique, is its most populous province (5,758,920 inhabitants), and contains both vast rural areas and the country’s third-largest city. Sofala province (2,221,803 inhabitants) is in Mozambique’s central region. Research will be conducted in 12 districts (four assigned to SBIRT-CTS and eight to mSBIRT), which constitute approximately 60% of the provinces’ populations (i.e., 4,788,430 people). Every district comprises clinics with diverse degrees of urbanicity, including clinics that are rural, suburban, and urban. In both provinces, mental health services are presently provided only at district psychiatric clinics by one to two psychiatric technicians per district and one to five psychologists per province.
Each district has approximately seven primary care clinics staffed by two to five primary care providers. Approximately five CHWs per clinic attend to 250–400 families in their community annually, depending on each family’s health needs. The CHWs are the first level of contact in the community and are part of the National Mozambique Health System. They are involved in testing for and treatment of infectious disease (malaria, HIV, and TB) and in maternal-child health services.
Study Procedures
Overview.
Year 1 will be devoted to SBIRT implementation (32), which will involve training and supervising staff, monitoring services and fidelity, providing implementation feedback to providers, and interviewing providers about implementation. In year 2, we will assess the sustainability of mSBIRT and SBIRT-CTS delivery (32). These implementation and sustainability phases will occur within the context of the cluster-randomized trial. In year 3—the scale-up phase—the more cost-effective strategy will be implemented in districts that received the less effective strategy.
Regardless of the trial arm, Ministry of Health procedures require that individuals who are reached, in need, and provided with care be seen weekly and screened with SBIRT. Those who are reached and engaged in follow-up constitute a retained population. Each weekly contact with a provider is documented by the first two items from the AUDIT, the 12-Item Short Form Health Survey (SF-12) (33), and the Mental Wellness Tool (mwTool, described below).
All patients and providers will provide written informed consent. Even though risks to provider participants are minimal, patient participants may experience increased distress and suicidal ideation or report physical or sexual abuse. In accordance with the Ministry of Health safety procedures, local coinvestigators (all clinicians) will be notified of an acute crisis within 24 hours. Additionally, we will convene a five-member data safety and monitoring board that will include an implementation scientist, statistician, mental health researcher with expertise in low-resource settings, Mozambique context expert, and ethicist. They will assist in monitoring adverse events and meet annually.
Implementation phase.
In the initial 6 months of the implementation phase, SBIRT and motivational interviewing experts will provide a 1-week training for Ministry Mental Health specialists. Trainees will present five cases in a group modality to become certified national trainers and supervisors. These newly minted trainers will then train and supervise the CHWs. We, the research team, will work with the trainers to adapt and pilot mSBIRT. We will also conduct a mixed-methods evaluation of the acceptability and feasibility of tablet use and capture CHWs’ intentions of using mSBIRT.
As part of the larger mental health study (31), all CHWs will be trained to use a tablet-based, comprehensive mental disorders assessment tool—the mwTool—that identifies presence of substance use disorders, common mental disorders, severe mental disorders, and acute risk for suicide. We will measure trainees’ knowledge (information), attitudes toward people with hazardous drinking (motivation), and self-efficacy (skills) in providing SBIRT before and after training (25, 29).
During the following 6 months of the implementation phase, the research team will provide active supervision through observation of role-play and case presentation sessions. CHWs will complete three cases and submit transcripts of the case sessions to their supervisor. Supervisors will evaluate the transcripts by using a treatment fidelity checklist comprising core intervention components and provide feedback to CHWs. If supervisors determine that a CHW conducts the SBIRT with insufficient fidelity or lacks competency, supervision and clinical support will continue until fidelity and competency are achieved.
The mSBIRT application software guides providers in following standardized treatment steps involving interactive methods, including knowledge tabs with tips and sample scripts, and automated processes to enable providers to recognize any clinical “red flags.” Metadata from the application will track individual provider implementation outcomes (adoption) and fidelity. Provider-level user data will indicate patients seen, key activity completion, and sessions per patient. If the user data of any provider appear to deviate from the standard sequence of activities, this will be flagged for supervisor intervention and corrective feedback to maximize fidelity. We will capture similar data in the SBIRT-CTS via provider and supervision checklists.
Sustainability phase.
The 12-month sustainability phase will begin once all CHWs and supervisors have achieved competency in either mSBIRT or SBIRT-CTS. We will monitor the CHWs, supervisors, and any Ministry of Health personnel involved in the study, but we will not provide supervision unless requested or deemed necessary on the basis of performance. We will sample six clinics in each arm from rural (N=2), suburban (N=2), and urban (N=2) areas. We will conduct 12 audio-recorded focus groups (1–1.5 hours each) with CHWs (one group per catchment area) and 24 semistructured household interviews with patients or their family members (two interviews per catchment area).
Scale-up phase.
In the scale-up phase, interviews will continue only in the six clinics of the superior arm: one focus group with CHWs at each clinic (N=6) and two household interviews per catchment area (N=12).
Screening and treatment.
The goal is for CHWs to use the mwTool to screen annually every household in their catchment area. For those who score positive for substance use disorders, CHWs will administer the AUDIT whose score will guide intervention. Patients with an AUDIT score of abstinence or low risk (<5) will receive feedback and follow-up in 4 weeks. Those with scores 5–15 will be considered to be at risk for or to have hazardous substance use and will receive a brief intervention weekly for 4 weeks. Patients who score in the harmful use range (AUDIT score ≥16) or in the dependent range (≥20) will also receive the four-session intervention plus medical monitoring by a primary care provider or psychiatric technician. Before the fourth session, CHWs will administer the AUDIT again. The fourth session reassessment will determine whether patients will be discharged (if they have an AUDIT score indicating abstinence or low risk), referred to an ongoing group intervention at the community level (AUDIT score is the same or improved, but not in low-risk range), or treated by a primary care provider or psychiatric technician (if they have worsening symptoms).
SBIRT sessions last 45–60 minutes and involve screening and a brief intervention consisting of providing feedback and health information, engaging patients in motivational conversations, and negotiating a plan for change (34). SBIRT incorporates principles of motivational interviewing—using an empathic style, supporting motivation and self-efficacy for change (35), building awareness about effects of alcohol use, and creating an action plan. We will establish a Mozambican SBIRT-mSBIRT workgroup with specialists who have expertise treating substance use disorders to adapt the treatment to the Mozambican context. Our adaptation workgroup will provide input into the mSBIRT application itself and suggestions for the general content and icon development.
Measures.
To determine the effectiveness and implementation outcomes, we will use the AUDIT and the mwTool. In both arms, the CHWs will screen household members yearly for mental health and substance use problems with the mwTool, which incorporates some AUDIT items as well as screeners for other mental health problems. Each CHW has a catchment area of 250–400 households (36). To decrease CHW burden, the CHWs will administer the mwTool to only two household members (ages ≥16 years) per home but will ask about other household members. Local research has shown that surrogate respondents provide valid information on alcohol intake (8). CHWs will individually administer the full AUDIT (37) and SF-12 (33, 38–40) to anyone age ≥16 years screening positive for substance use disorders on the mwTool (Table 2). On the basis of AUDIT scores, CHWs will deliver either mSBIRT or SBIRT-CTS, according to the trial arm. The full AUDIT will be administered at sessions 1 and 4 to measure effectiveness. The study measures are organized by RE-AIM outcomes (Table 2) (41–55).
| Domain | Definition | Provider |
|---|---|---|
| Implementation impact outcomes | ||
| Reachb | Those who access care or initiate SBIRT among individuals who screen positive on the mwTool (score for men ≥4, women ≥3 [41–43]), indicating a health care need. Characteristics of those reached vs. not reached (including gender, age, health outcomes, urbanicity).b | Community health worker (CHW) |
| Adoption at clinic and staff level | Number of clinics or CHWs using SBIRT (numerator) over those expected to use SBIRT (denominator, all in each district).c | Research assistant (RA) and CHW |
| Clinics | Characteristics of clinics adopting SBIRT vs. not adopting in both conditions (rural or urban, number of CHWs, patient characteristics such as comorbid conditions, access barriers), Evidence-Based Practice Attitudes Scale (15 items [44]), Ready Set Change (organizational readiness for change) (66), RE-AIM Planning Tool (27-item scale [26, 45]), and Program Sustainability Assessment Tool (40 items [46, 47]). | RA, CHW, or supervisor |
| Staff (predictors of SBIRT implementation [35, 48]) | Characteristics of CHWs adopting vs. not adopting in both conditions (age, gender, work, [49–51])b; intentions (9 items and 7-point scale), asking how willing and how likely one is to use each SBIRT component (52, 53); attitudes toward each SBIRT component; norms or perception that other people like them will use the SBIRT (6 standard item stems); and self-efficacy (6 statements) measuring CHWs’ and supervisors’ perceptions of their skills and abilities to perform SBIRT. | |
| Implementation fidelityc | Consistency of implementation across time in both study arms. Characteristics of clinics and CHWs as described in adoption. | RA and CHW |
| Maintenance | Used over a 2-year period. Number of clinics using SBIRTc; adaptations to protocolsc; characteristics of clinics and CHWs as described in adoption.c | RA and CHW |
| Clinical effectiveness outcomes | ||
| Symptomsb | Assessed with AUDIT (41–43, 55), a 10-item instrument developed by the World Health Organization. | CHW and patient |
| Functioningb | Assessed with 12-Item Short Form Health Survey (SF-12) (38–40). | CHW and patient |
| Other health-related outcomesb | Assessed through electronic medical records (e.g., HIV viral load, adherence to care for HIV or tuberculosis). | CHW and patient |
| Cost-effectiveness outcomes | ||
| Cost (effort and time)c | Assessed with Stages of Implementation Completion Scale (SIC); readiness or preimplementation, adoption or implementation, and sustainability and competency (32); and Cost of Implementing New Strategies (COINS; mapping implementation resources using the SIC) (69). | RA and CHW |
TABLE 2. Reach, Effectiveness, Adoption, Implementation, Maintenance (RE-AIM) measures for mixed-methods data collectiona
For aim 3, we will use a mixed-methods approach to identify organizational and clinician factors that affect SBIRT implementation. Interview guides and analyses will be guided by our conceptual model combining the theory of planned behavior with organizational constructs from the CFIR (28). We will triangulate quantitative and qualitative data to identify the factors influencing adoption and sustainability and facilitate translation of research findings into effective policy. In the “crossover” phase, adoption and causal model measures will be administered to all participants. In each focus group, we will present our quantitative results in easy-to-interpret graphics and descriptions. We will ask participants to consider the results, adoption rate, and any associations between clinician (intentions) and organizational (culture, climate) factors and adoption.
Analysis Strategy
Hypothesis testing.
The cluster-randomized trial will address hypotheses 1 and 2 regarding the implementation and effectiveness of the interventions across the eight mSBIRT and four SBIRT-CTS districts. The hypotheses will be used to compare the interventions’ effect on outcomes measured for individuals and CHWs aggregated at the clinic level. We expect to include 62 clinics across the 12 districts, employing 417 CHWs who serve between 104,250 and 166,800 households. Clinic-level outcomes will be measured at baseline and then at 12, 18, and 24 months. Because clinics are nested within districts, we will use hierarchical linear models (HLMs), which will include random effects on each level and fixed effects for the intervention indicator and for the time variable.
Hypothesis 1 will test whether mSBIRT is superior to SBIRT-CTS on each of the RE-AIM implementation outcomes, by using HLM analysis, including an indicator for intervention, time (baseline, 12, 18, and 24 months), and intervention × time interaction. The magnitude and statistical significance of the interaction term will indicate whether a change in implementation outcomes over time significantly differs by intervention arm. If the arms differ on baseline measures, we will include those measures as control variables in the HLM.
Hypothesis 2 investigates the interventions’ effect on clinical outcomes (first and second subhypotheses) and on costs (third subhypothesis). For the first subhypothesis, we will use an HLM (controlling for intervention and time) to estimate changes in hazardous drinking and health-related quality of life at months 12, 18, and 24 after SBIRT exposure. Overall intervention effects will be tested over time, and interactions of the intervention by gender, age, and urbanicity of the patient will test for effect modification. For the second subhypothesis, we will again use an HLM (controlling for intervention and time) to predict changes in scores from baseline in health-related outcomes (viral load and HIV or TB care adherence) at months 12, 18, and 24 by using changes in hazardous drinking from baseline. We will assess effect modification by gender, age, and urbanicity by including them as interaction terms. Finally, the third subhypothesis will test whether mSBIRT is more cost-effective than SBIRT-CTS in improving both implementation (hypothesis 1) and clinical outcomes (first and second subhypotheses of hypothesis 2). Comparison of costs will be aggregated across the sustainability phase in year 2. We will assess health-related quality of life with the SF-12, which can be converted to construct quality-adjusted life years (QALYs) (56). QALY is a measure of health-related quality of life that incorporates estimates of mortality and morbidity rates into a single index for ease of comparison across conditions and interventions. Incremental cost-effectiveness ratios will identify the superior arm for the scale-up phase (numerator=difference in mean costs between arms, denominator=difference in reach and reduction in hazardous drinking). Uncertainty in costs and outcomes will be assessed with cost-effectiveness acceptability curves (57–59).
Power analysis for hypotheses 1 and 2.
Power was calculated on the basis of participation of approximately 480 CHWs located across clinics in 12 districts and an anticipated dropout rate of about 20%. Each CHW visits between 250 and 400 families in the community at least once a year, resulting in a coverage of approximately 150,000 families (about 75,000 families within each randomized condition). For hypothesis 1, to test for differences in implementation outcomes between randomized study condition at the family level (e.g., percentage households screened), we will have 80% power to detect small to moderate effect sizes between the two conditions, with Cohen’s d=0.23–0.36, following standard power calculations for cluster-randomized trials (60). This power calculation was derived by assuming an intraclass correlation (ICC) of families within the 12 randomized districts in the typical range for health care outcomes across randomized regions, that is, an ICC of 0.02–0.05 (61, 62). For hypothesis 2, we conservatively assumed that 3%−5% of the population will be identified as having engaged in hazardous drinking and will be enrolled in either intervention. Thus, we expect that 4,500–7,500 clients will enroll across the two randomized study arms nested within 480 CHWs who will provide treatment. Given this sample of treated individuals, we will have 80% power to detect intervention differences of Cohen’s d ≥0.16. This power was calculated by assuming an ICC of 0.4 for client outcomes within CHWs, which is conservatively at the high end of ICCs found within health service providers in low-income settings (63). All power calculations assume an alpha of 0.05 and that all tests are two-tailed.
For hypothesis 3, we will examine organizational and clinician-level factors affecting implementation of the intervention with data from the implementation, sustainability, and scale-up phases. The first subhypothesis of hypothesis 3 is that organizational factors such as urbanicity, culture, and climate will affect clinicians’ attitudes, perceived norms, and self-efficacy regarding SBIRT use, which will in turn affect their intentions to use SBIRT. The second hypothesis is that clinicians’ demographic characteristics and intentions to use SBIRT will affect SBIRT adoption and use. To test both subhypotheses, we will use structural equation modeling (64, 65) to estimate the effect of organizational factors and clinicians’ demographic characteristics on determinants of intentions (attitudes, norms, and self-efficacy) and the effect on intentions (Figure 1). Organizational factors will include urbanicity, number of staff, patients’ characteristics and the ready-set-change measure (66). Consistent with best practice, we will aggregate these organizational measures at the clinic level. Intentions and determinants of intentions to adopt SBIRT will be measured with validated self-report measures (Table 2). The use of structural equation modeling allows direct incorporation of clinic-level random effects and provides straightforward estimation of indirect effects. We will also use structural equation modeling to estimate the effect of intentions on adoption, while controlling for the clinic and clinician characteristics described above. We will use interaction terms to further test for effect modification of the organizational factors on the association between intentions and adoption.
For qualitative analysis, interviews and focus groups will be digitally recorded, transcribed, and loaded into Nvivo software for analysis. To avoid bias, analysts will be blinded to implementation outcomes. Initial coding will draw on the CFIR, and an inductive process of iterative coding will identify themes and categories. Analysts will then be unblinded to implementation outcomes to determine distinguishing constructs. Finally, for mixed-methods data analysis, quantitative data are gathered before qualitative data and weighted equally: QUAN→QUAL (67, 68); the function is complementary, and the process is connecting (having the qualitative data set build on the quantitative data set) (67).
Results
The more cost-effective strategy will be scaled up in the other districts by using procedures outlined in the implementation phase. However, during the scale-up phase, trained Ministry of Health personnel will be charged with delivery, training, and supervision of the superior strategy in all clinics without research team support. The scale-up phase is critical to identify facilitators and barriers through process evaluation and continued tracking of internal and external factors in clinics that started in the superior arm and those that switch strategies.
Next Steps
We will work with stakeholders from multiple sectors to use the lessons learned from our study to develop an implementation tool kit that can guide SBIRT for community-wide HDS scale-up and dissemination in LMIC countries, New York State, and city departments of health and mental health. We will review the data, create a draft tool kit, and present it to LMIC policy makers and WHO representatives. Policy maker feedback will help finalize the tool kit. This kit will be freely available to all research collaborators.
1 : Alcohol: a global health priority. Lancet 2009; 373:2173–2174Crossref, Medline, Google Scholar
2 : Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet 2009; 373:2223–2233Crossref, Medline, Google Scholar
3
4 : Steps towards constructing a global comparative risk analysis for alcohol consumption: determining indicators and empirical weights for patterns of drinking, deciding about theoretical minimum, and dealing with different consequences. Eur Addict Res 2001; 7:138–147Crossref, Medline, Google Scholar
5 : Alcohol as a key area. BMJ 1991; 303:766–769Crossref, Medline, Google Scholar
6 : The global distribution of average volume of alcohol consumption and patterns of drinking. Eur Addict Res 2003; 9:147–156Crossref, Medline, Google Scholar
7 : Alcohol consumption in Mozambique: regular consumption, weekly pattern and binge drinking. Drug Alcohol Depend 2011; 115:87–93Crossref, Medline, Google Scholar
8 : Alcohol consumption in Mozambique: results from a national survey including primary and surrogate respondents. Ann Hum Biol 2012; 39:534–537Crossref, Medline, Google Scholar
9 : Depression among female heads-of-household in rural Mozambique: a cross-sectional population-based survey. J Affect Disord 2018; 227:48–55Crossref, Medline, Google Scholar
10 Inquérito de Indicadores de Imunização, Malária e HIV/SIDA em Moçambique (IMASIDA) 2015. Maputo, Moçambique, Ministério da Saúde (MISAU) Instituto Nacional de Estatística (INE), 2018. http://dhsprogram.com/pubs/pdf/AIS12/AIS12.pdfGoogle Scholar
11 : Alcohol use and antiretroviral adherence: review and meta-analysis. J Acquir Immune Defic Syndr 2009; 52:180–202Crossref, Medline, Google Scholar
12 : Alcohol consumption and infectious diseases in South Africa. Lancet 2009; 374:2053Crossref, Medline, Google Scholar
13 : Evidence-based guidelines for mental, neurological, and substance use disorders in low- and middle-income countries: summary of WHO recommendations. PLoS Med 2011; 8:e1001122Crossref, Medline, Google Scholar
14 mhGAP Mental Health Gap Action Programme: Scaling Up Care for Mental, Neurological and Substance Use Disorders. Geneva, World Health Organization, 2008. https://www.who.int/mental_health/mhgap_final_english.pdfGoogle Scholar
15 : Brief Intervention for Hazardous and Harmful Drinking: A Manual for Use in Primary Care. Geneva, World Health Organization, 2001. https://apps.who.int/iris/bitstream/handle/10665/67210/WHO_MSD_MSB_01.6b.pdf;jsessionid=78DBD725F26E7CB4FEB8134BCCD4432F?sequence=1. Accessed March 26, 2021Google Scholar
16 : Effectiveness of brief interventions in primary health care settings to decrease alcohol consumption by adult non-dependent drinkers: a systematic review of systematic reviews. Prev Med 2015; 76(suppl):S33–S38Crossref, Medline, Google Scholar
17 : Screening, brief intervention and referral to treatment (SBIRT): implementation barriers, facilitators and model migration. Addiction 2017; 112(suppl 2):23–33Crossref, Medline, Google Scholar
18 : Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption—II. Addiction 1993; 88:791–804Crossref, Medline, Google Scholar
19
20 : The promises and pitfalls of digital technology in its application to alcohol treatment. Alcohol Res 2014; 36:131–142Medline, Google Scholar
21 : Point-of-care mobile application to guide healthcare professionals in conducting substance use screening and intervention: a mixed-methods user experience study. ACI Open 2019; 3:e1–e12. doi: 10.1055/s-0039-1684002Crossref, Google Scholar
22 : Mobile technology-based interventions for adult users of alcohol: a systematic review of the literature. Addict Behav 2016; 62:25–34Crossref, Medline, Google Scholar
23 : Mobile health (mHealth) approaches and lessons for increased performance and retention of community health workers in low- and middle-income countries: a review. J Med Internet Res 2013; 15:e17Crossref, Medline, Google Scholar
24 : Contributions of mobile technologies to addiction research. Dialogues Clin Neurosci 2016; 18:213–221Crossref, Medline, Google Scholar
25 : The theory of planned behavior. Org Behav Hum Dec Proc 1991; 50:179–211. doi: 10.1016/0749-5978(91)90020-TCrossref, Google Scholar
26 : Applying the RE-AIM framework to assess the public health impact of policy change. Ann Behav Med 2007; 34:105–114Crossref, Medline, Google Scholar
27 : Evaluating the public health impact of health promotion interventions: The RE-AIM Framework. Am J Public Health 1999; 89:1222–1327Crossref, Medline, Google Scholar
28 : Fostering implementation of health services research findings into practice: a consolidated framework for advancing implementation science. Implement Sci 2009; 4:50Crossref, Medline, Google Scholar
29
30 : Effectiveness-implementation hybrid designs: combining elements of clinical effectiveness and implementation research to enhance public health impact. Med Care 2012; 50:217–226Crossref, Medline, Google Scholar
31 : Partnerships in Research to Implement and Disseminate Sustainable and Scalable Evidence-Based Practices (PRIDE) in Mozambique. Psych Serv (Epub Dec 18, 2020)Link, Google Scholar
32 : Observational measure of implementation progress in community based settings: the Stages of Implementation Completion (SIC). Implement Sci 2011; 6:116Crossref, Medline, Google Scholar
33 : The estimation of a preference-based measure of health from the SF-12. Med Care 2004; 42:851–859Crossref, Medline, Google Scholar
34 : Project ASSERT: an ED-based intervention to increase access to primary care, preventive services, and the substance abuse treatment system. Ann Emerg Med 1997; 30:181–189Crossref, Medline, Google Scholar
35 : Understanding Attitudes and Predicting Social Behavior. Englewood Cliffs, NJ, Prentice-Hall, 1980Google Scholar
36 :
37 : Depression, alcohol use and adherence to antiretroviral therapy in sub-Saharan Africa: a systematic review. AIDS Behav 2012; 16:2101–2118Crossref, Medline, Google Scholar
38 : Assessing health and well-being among older people in rural South Africa. Glob Health Action 2010; 3(Supp. 2):23–35Crossref, Google Scholar
39 : Cross-cultural adaptation of health-related quality of life measures: literature review and proposed guidelines. J Clin Epidemiol 1993; 46:1417–1432Crossref, Medline, Google Scholar
40
41 : AUDIT: The Alcohol Use Disorders Identification Test Guidelines for Use in Primary Care. Geneva, World Health Organization, 2001Google Scholar
42 : The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Ambulatory Care Quality Improvement Project (ACQUIP). Alcohol Use Disorders Identification Test. Arch Intern Med 1998; 158:1789–1795Crossref, Medline, Google Scholar
43 :
44 : Mental health provider attitudes toward adoption of evidence-based practice: the Evidence-Based Practice Attitude Scale (EBPAS). Ment Health Serv Res 2004; 6:61–74Crossref, Medline, Google Scholar
45 : What does it mean to “employ” the RE-AIM model? Eval Health Prof 2013; 36:44–66Crossref, Medline, Google Scholar
46 : The Program Sustainability Assessment Tool: a new instrument for public health programs. Prev Chronic Dis 2014; 11:130184Crossref, Medline, Google Scholar
47 : Using the Program Sustainability Assessment Tool to assess and plan for sustainability. Prev Chronic Dis 2014; 11:130185Crossref, Medline, Google Scholar
48 : A reasoned action approach to health promotion. Med Decis Making 2008; 28:834–844Crossref, Medline, Google Scholar
49 : Autoefficacia Percepita Nella Ricerca del Lavoro: Scale di Misura. Isfol, Bisogni, Valori e Autoeficacia Nella Scelta del Lavoro. Roma, ISFOL, 2007, pp 133–142Google Scholar
50 : Occupational Self-Efficacy Scale-Short Form (OSS-SF): adaptation and evidence of construct validity of the Brazilian version. Rev Bras Orientaç Prof 2014; 15:65–74Google Scholar
51 : Work Self-Efficacy Scale and Search for Work Self-Efficacy Scale: a validation study in Spanish and Italian cultural contexts. Rev Psicol Trab Organ 2010; 26:201–210. doi: 10.5093/tr2010v26n3a4Crossref, Google Scholar
52 : Predicting and Changing Behavior: The Reasoned Action Approach. New York, Psychology Press, 2010Google Scholar
53 : Differentiating between precursor and control variables when analyzing reasoned action theories. AIDS Behav 2010; 14:225–236Crossref, Medline, Google Scholar
54 : The AB scale: an operational definition of belief and attitude. Hum Relat 1962; 15:35–44. doi: 10.1177/001872676201500104Crossref, Google Scholar
55 : Men who have sex with men in Mozambique: identifying a hidden population at high-risk for HIV. AIDS Behav 2015; 19:393–404Crossref, Medline, Google Scholar
56 : The estimation of a preference-based measure of health from the SF-36. J Health Econ 2002; 21:271–292Crossref, Medline, Google Scholar
57 : Decision Modelling for Health Economic Evaluation (Handbooks in Health Economic Evaluation). New York, Oxford University Press, 2006Google Scholar
58 : Methods for the Economic Evaluation of Health Care Programmes. New York, Oxford University Press, 2015Google Scholar
59 : Cost-Effectiveness in Health and Medicine. New York, Oxford University Press, 2016Crossref, Google Scholar
60 : Sample size calculations for cluster randomised controlled trials with a fixed number of clusters. BMC Med Res Methodol 2011; 11:102Crossref, Medline, Google Scholar
61 : Intraclass correlation coefficients for cluster randomized trials in care pathways and usual care: hospital treatment for heart failure. BMC Health Serv Res 2014; 14:84Crossref, Medline, Google Scholar
62 : Comparison of methods for estimating the intraclass correlation coefficient for binary responses in cancer prevention cluster randomized trials. Contemp Clin Trials 2012; 33:869–880Crossref, Medline, Google Scholar
63 Exploring sources of variability in adherence to guidelines across hospitals in low-income settings: a multi-level analysis of a cross-sectional survey of 22 hospitals. Implement Sci 2015; 10:60Crossref, Medline, Google Scholar
64 : The quantitative analysis of reasoned action theory. Ann Am Acad Pol Soc Sci 2012; 640:28–41 doi: 10.1177/0002716211424265Crossref, Google Scholar
65 : Using the Integrative Model to explain how exposure to sexual media content influences adolescent sexual behavior. Health Educ Behav 2011; 38:530–540Crossref, Medline, Google Scholar
66 : Ready, set, change! Development and usability testing of an online readiness for change decision support tool for healthcare organizations. BMC Med Inform Decis Mak 2016; 16:24Crossref, Medline, Google Scholar
67 : Mixed method designs in implementation research. Adm Policy Ment Health 2011; 38:44–53Crossref, Medline, Google Scholar
68 : Implementation of evidence-based practice in child welfare: service provider perspectives. Adm Policy Ment Health 2007; 34:411–419Crossref, Medline, Google Scholar
69 : The Cost of Implementing New Strategies (COINS): a method for mapping implementation resources using the stages of implementation completion. Child Youth Serv Rev 2014; 39:177–182Crossref, Medline, Google Scholar



